Ion exchange chromatography overview скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Ion exchange chromatography overview в качестве 4k
У нас вы можете посмотреть бесплатно Ion exchange chromatography overview или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Ion exchange chromatography overview в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Ion exchange chromatography overview
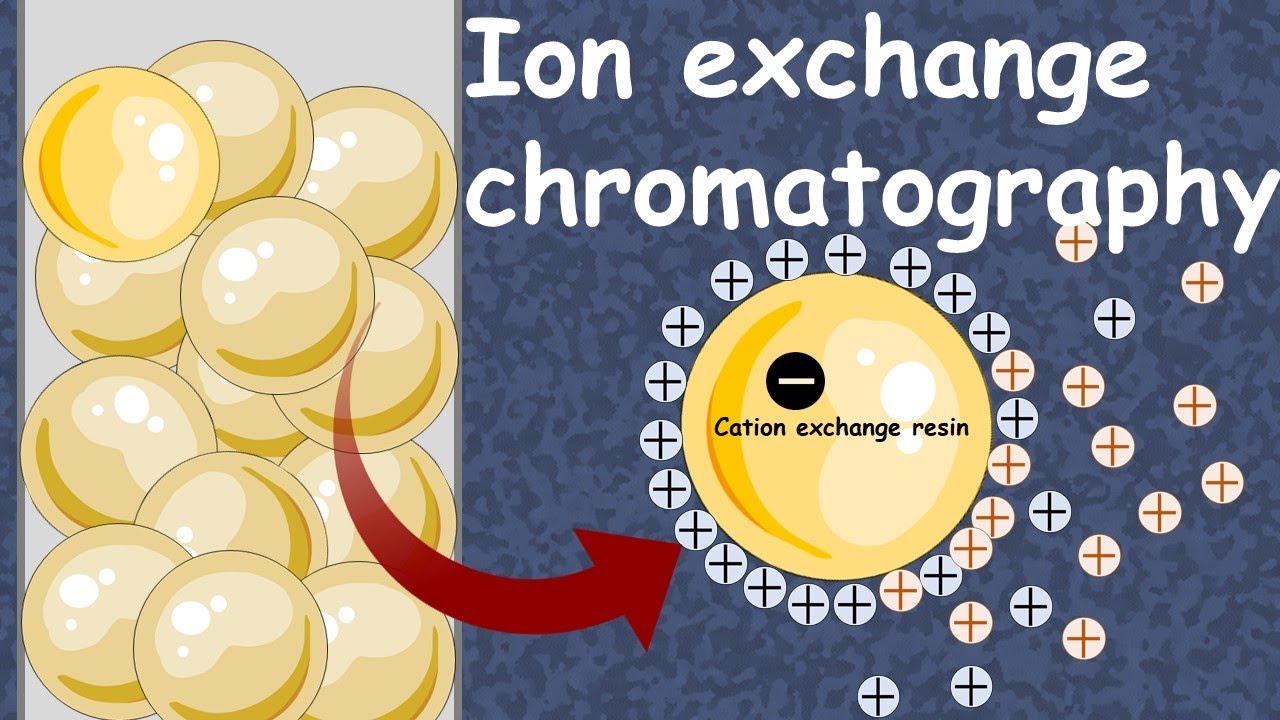
Ion exchange chromatography is one of the main methods we use to separate proteins. It separates them based on charge. The terms can get confusing so here’s a summary & then some more details: Cation exchange Your protein is cationic (positively-charged) & the resin is negatively-charged Good for “basic” proteins - proteins with a high pI (due to lots of Arg, Lys, &/or His) You need to be at a pH below the pI to ensure your protein is positively-charged Anion exchange Your protein is anionic (negatively-charged) & the resin is positively-charged Good for “acidic” proteins - proteins with a low pI (due to lots of Glu &/or Asp) You need to be at a pH above the pI to ensure your protein is negatively-charged In either case, you can add high salt to exchange salt ions for your protein on the resin, allowing your protein to un-stick from the resin (elute). Now some more detail below & much more here: blog form: http://bit.ly/ionexchangechromatography ; YouTube: • Ion exchange chromatography protein purifi... Amino acids all have the same generic backbone that they use to link together & then they have unique “side chains” aka “side groups” aka “R groups” The generic backbone part has an amino (-NH₂) group on 1 end & a carboxyl -(C=O)-OH group on the other end. They link up amino to carboxyl in an amide aka peptide bond —NH-(C=O)— , shed a water in the process & the part that’s left we often refer to as the “residue” - so when alanine is by itself, we call it a free amino acid. But when it’s in a chain (peptide) we call it a residue. When you have free amino acids, they all have at least 2 groups that can give or take hydrogens. The amino end can exist as NH₂ (neutral) or -NH₃⁺ (+ charged) & the carboxyl group can exist as -(C=O)-OH (neutral) or -(C=O)-O⁻ ( charged). So a free amino acid can be +, -, or neutral. The pKas of these groups depend on the amino acid but are typically ~10 & ~2 respectively. Physiological pH (the pH inside your body) is about 7.4, which is above 2, so most of the carboxyl groups are protonated & thus in the negatively-charged carboxylate form. But below 10, so most of the amino ends are in the +-charged NH₃⁺ form. So the charges cancel out – we call this a ZWITTERION When you link amino acids together, you’re only left with 1 amino end & one carboxyl end. So you only have to take those group’s protonation state into account once. But some of the side chains in the residues in between those ends can get protonated or deprotonated as well & when we talk about pKa’s for amino acids in the context of proteins or peptides, we usually are talking about the pKa of the side chain (if there is one). Sometimes this is referred to as the pKR or pK3. There are 2 side chains that are frequently deprotonated at physiological pH - we call these ACIDIC because they donate H⁺s & when they do they become negatively charged & now capable of accepting H⁺s (acting as a base) so we call them “conjugate bases.” It can seem kinda confusing because “acidic” residues often play important roles by acting as bases in their deprotonated form. The “acidic” refers to its NEUTRAL form being acidic. These side chains are: Aspartic acid (Asp, D)(pKa ~3.65), which gives up a H⁺ to become aspartate & Glutamic acid (Glu, E)(pKa ~4.25) which gives up a H⁺ to become glutamate. Cysteine (Cys, C) & tyrosine (Tyr, Y) can can also deprotonate to give negatively charged chains, but they do so much less readily - pKa of 8.4 for Cys & 10.5 for Tyr. There are 3 side chains that are frequently protonated at physiological pH - we call these “basic.” Lysine (Lys, K) (pKa ~ 10.28) & Arginine (Arg, R) (pKa ~13.2) are predominantly protonated at cellular pH, but Histidine (His, H) (5.97) is more “iffy” (remember that pKa tells you when HALF the groups are deprotonated on average so it’s not like you hit the pKa & bam they’re all deprotonated - you have a mix. Also, an important caveat is that pKas are context-dependent so the pKa you get from a table is likely close to but not exactly the “real” pKa in the situation you’re looking at. We can calculate the pI (isoelectric point), that pH at which the protein is neutral overall. It doesn’t mean that each individual amino acid is neutral, just that the non-neutral ones balance out. The higher the pI, the more H⁺-rich the protein is likely to be ➔ you have to take it to a higher pH (more basic meaning less free H⁺) before it will shed H⁺s. We often refer to such proteins that are + charged at neutral pH as “basic” & they get that “basicity” because they have lots of basic residues (His, Lys, &/or Arg). They’re great for binding negatively-charged things like DNA or RNA. Proteins with pI’s below neutral are “acidic” and they usually have lots of Glu’s and Asp’s. finished in comments