Apoptosis Explained: p53 Activation, Cytochrome c Release, the Apoptosome, and the Caspase Cascade скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Apoptosis Explained: p53 Activation, Cytochrome c Release, the Apoptosome, and the Caspase Cascade в качестве 4k
У нас вы можете посмотреть бесплатно Apoptosis Explained: p53 Activation, Cytochrome c Release, the Apoptosome, and the Caspase Cascade или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Apoptosis Explained: p53 Activation, Cytochrome c Release, the Apoptosome, and the Caspase Cascade в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Apoptosis Explained: p53 Activation, Cytochrome c Release, the Apoptosome, and the Caspase Cascade
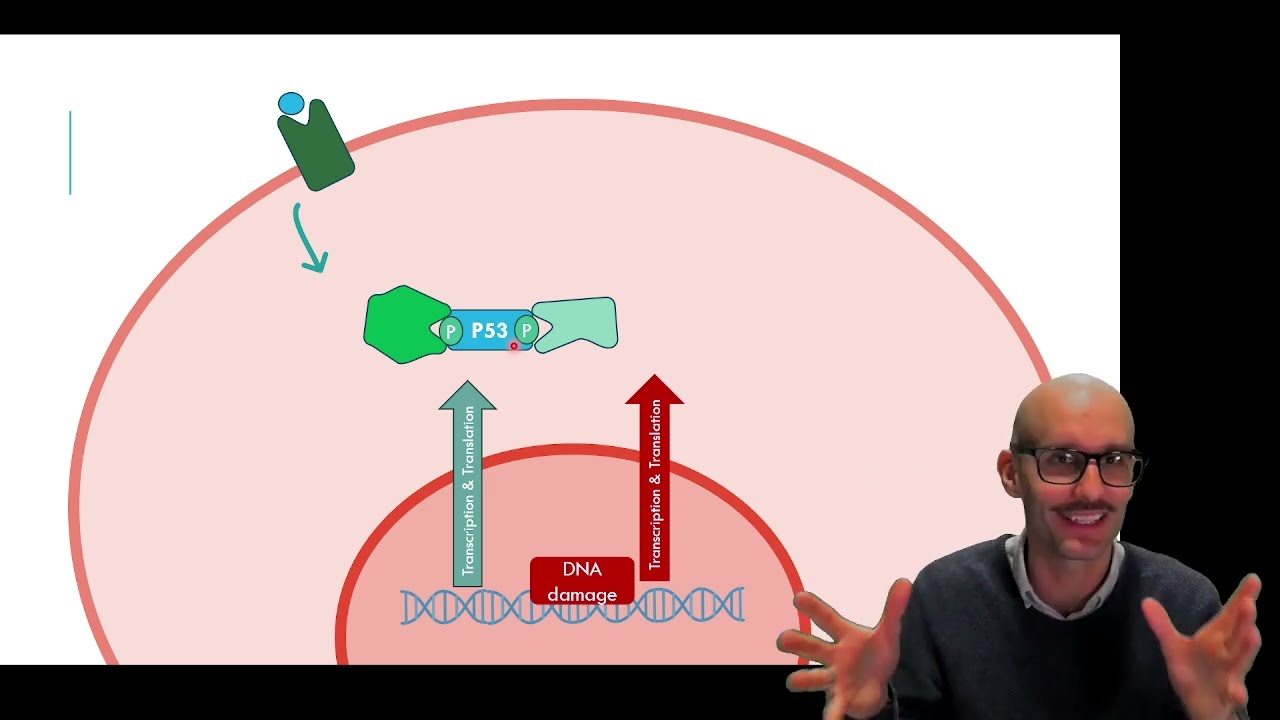
This video walks through the molecular sequence that drives apoptosis (programmed cell death), starting with the key regulator p53. Under normal conditions, p53 is continuously produced but kept low because it is ubiquitinated and degraded by the proteasome. When a cell receives stress signals, either intrinsic (for example DNA damage) or extrinsic (death receptor signalling), kinases phosphorylate p53. This phosphorylation changes p53’s shape, prevents its ubiquitination, and allows p53 to accumulate and enter the nucleus to switch on pro-apoptotic genes. A major downstream step is the production of pore-forming proteins that permeabilise the mitochondrial membrane. This releases cytochrome c (and adaptor proteins) into the cytosol, where they assemble into the apoptosome (described as a “ninja star” complex). The apoptosome recruits inactive caspase zymogens and activates them through proximity-driven cleavage, triggering a caspase activation cascade. This cascade amplifies the signal dramatically, activating enzymes that fragment DNA into manageable pieces and proteases that dismantle cellular proteins, including the cytoskeleton. The breakdown of the cytoskeleton, together with changes in ion and water movement, drives membrane blebbing and the packaging of cell contents into apoptotic bodies for clearance.