How will you separate U235 from natural Uranium? Nuclear Chemistry | Physical Chemistry скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: How will you separate U235 from natural Uranium? Nuclear Chemistry | Physical Chemistry в качестве 4k
У нас вы можете посмотреть бесплатно How will you separate U235 from natural Uranium? Nuclear Chemistry | Physical Chemistry или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон How will you separate U235 from natural Uranium? Nuclear Chemistry | Physical Chemistry в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
How will you separate U235 from natural Uranium? Nuclear Chemistry | Physical Chemistry
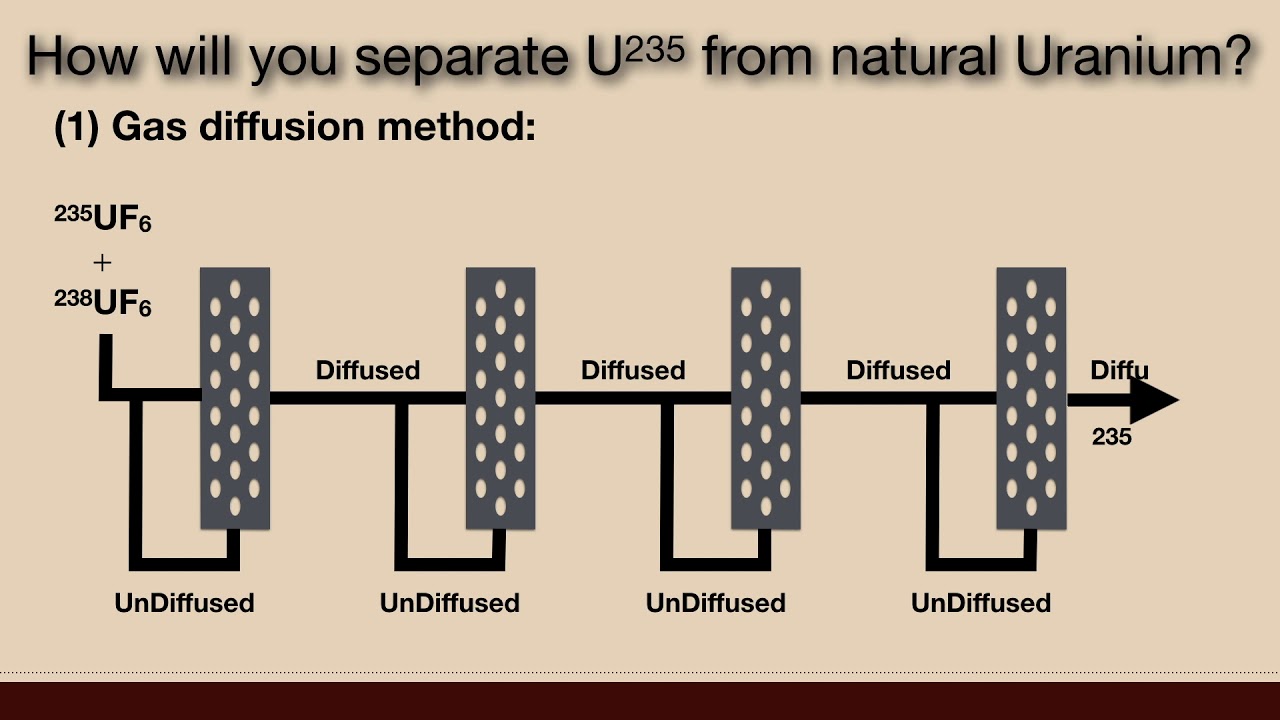
1. How will you separate U235 from natural Uranium? (OR) 2. Give an account of various methods used for separation of Isotopes? (OR) 3. What are the various methods used to separate isotopes? Describe any two in detail. Uranium is found in nature. It exists in two isotopic forms, which are 235U and 238U. The proportion of the two is 0.72% and 99.28% respectively. They can be separated by two methods. (1) Gas diffusion method: As the name suggests it is applicable only for gases. But uranium is in solid form, In this case, uranium is allowed to react with another element and the compound formed must be volatile. E.g. when U is allowed to react with F-atom they form Uranium hexafluoride, which is volatile, the method can now be applied. Now we have 235UF6, and 238UF6. After the separation of two isotopes, each isotope is defluorinated to obtain free Uranium. Principle: The method is based on Graham's Law of Diffusion. Grahams law of diffusion state that rate of diffusion of any gas is inversely proportional to the square root of its density i.e. r propto frac { 1 }{ sqrt { d } } where r is rate of diffusion and d is density. But d = frac { mass }{ volume } for constant volume we can say, therefore r propto frac { 1 }{ sqrt { mass } } or boxed { r propto frac { 1 }{ sqrt { Molecular Weight } } } It means light isotopes will be diffused more than the heavy isotopes. Working: The natural Uranium is allowed to combine with F, and form Uranium hexafluoride. The mixture of two. Uranium hexafluoride is allowed to pass through no. of diffusion boxes which contain porous porcelain pipes are used. These no. of the boxes are arranged one after the another on the common principal axis. With the help of a pump, we pass the mixture through the Box No.1. Here the diffusion of two isotopes takes place. As 235UF6 is lighter than 238UF6 so it will be diffused more. The diffusate is allowed to pass through Box No.2 with the help of a pump and undiffused part is sent back for diffusion to Box No.1. The process is continued after passing through 4000 boxes, the diffusate will be 99.9% of 235UF6. The big problem in the method of the arrangement of 4000 boxes. CASCADE solved the above problem of cycling and recycling through just only three boxes and the method is known as the cascade method of diffusion of gases. The method is useful for the separation of isotopes of Ne (20, 21, 22), N(14, 15), O(16, 17, 18) etc. (2) Chemical Exchange Method: The chemical exchange method is based on the fact that though the different isotopes of the same element undergo the same chemical reactions, the rate of these reactions are different for different isotopes as there is a difference in their activation energies. Another important observation is that one isotope of an element in a particular compound will exchange with another isotope of the same element in another compound. Consider the exchange of 14N and 15N. If gaseous ammonia 15NH3 is made to flow in the countercurrent through a solution of 14NH4OH which trickles down the column packed with glass helices as the catalyst the following exchange is observed: 15NH3 (g) + 14NH4OH (aq) ⇄ 14NH3 (g) + 15NH4OH (aq) The equilibrium constant for the exchange reaction is 1.033 at 25°c. Thus, the rate of exchange from Left to Right is somewhat greater than from Right to Left. Thus the heavier isotope tends to concentrate in the solution at the bottom and lighter in the gaseous state at the top. Similarly, stable isotopes of 12C and 13C can be separated by the chemical exchange H12CN (g) + 13CN (aq) ⇄ H13CN (g) + 12CN (aq) The equilibrium constant is 1.026 at 25°C. Electrochemistry - Physical Chemistry • What are different types of Reversible Ele... Solid State - Physical Chemistry • Explain the following terms | Solid State ... Gaseous State - Physical Chemistry • Postulates of Kinetic Molecular Theory of ... Colloidal States - Physical Chemistry • What is Colloidal Solution? | Colloidal St... Stereochemistry - Organic Chemistry • Explain Configuration and Conformation | S... Nanomaterials - Engineering Chemistry • Compare top down with bottom up Processes ... Water and Its Treatment - Engineering Chemistry • Explain why hard water gives out a curd li... Electrochemistry - Engineering Chemistry • Distinguish between metallic and electroly... Environmental Studies • MCQ on Environmental Studies Part 8 Optics - Applied Physics • What are cartesian sign conventions for sp... For Details Visit http://cepekmedia.co.nf http://cepek.hol.es/ http://edmerls.66Ghz.com/ http://edmerls.tk/








![Как измеряют самые слабые силы во Вселенной? [Veritasium]](https://imager.clipsaver.ru/6lK2JVwajTw/max.jpg)








![Пожалуй, главное заблуждение об электричестве [Veritasium]](https://imager.clipsaver.ru/6Hv2GLtnf2c/max.jpg)

