Carboxylic Acid Derivatives | Reactivity and Nucleophilic Acyl Substitution | Organic Chemistry скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Carboxylic Acid Derivatives | Reactivity and Nucleophilic Acyl Substitution | Organic Chemistry в качестве 4k
У нас вы можете посмотреть бесплатно Carboxylic Acid Derivatives | Reactivity and Nucleophilic Acyl Substitution | Organic Chemistry или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Carboxylic Acid Derivatives | Reactivity and Nucleophilic Acyl Substitution | Organic Chemistry в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Carboxylic Acid Derivatives | Reactivity and Nucleophilic Acyl Substitution | Organic Chemistry
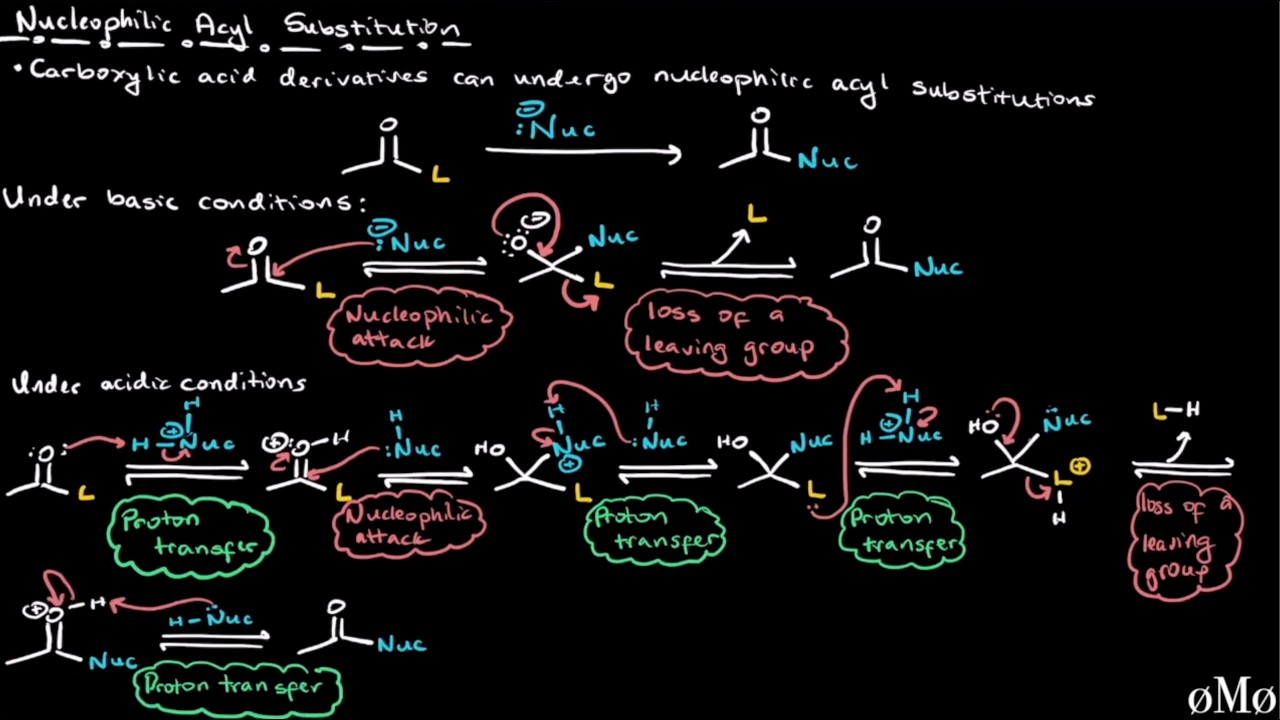
In this comprehensive organic chemistry video, we cover carboxylic acid derivatives. We explore the structural features that define these derivatives, including acid chlorides, anhydrides, esters, amides, and nitriles. You will learn how to rank their relative reactivity by analyzing inductive and resonance effects and how to propose reasonable mechanisms for nucleophilic acyl substitution reactions under various conditions. Key topics covered include: Identifying Derivatives: Understanding the oxidation state of the carbonyl carbon and how it defines carboxylic acid derivatives. Reactivity Trends: A deep dive into why acid chlorides are highly reactive due to induction, while amides are relatively unreactive due to strong resonance stabilization. Mechanism Breakdown: Step-by-step walkthroughs of nucleophilic acyl substitution, contrasting these reactions with the addition reactions seen in aldehydes and ketones. Condition-Based Mechanisms: Comparing reaction pathways under basic, neutral, and acidic conditions, emphasizing the role and timing of proton transfer steps. Timestamps: 00:00 Introduction to Carboxylic Acid Derivatives 00:12 Oxidation States and Identifying Derivatives 02:54 Ranking Relative Reactivity 04:01 Induction vs. Resonance: Case Study of Acid Chlorides and Amides 07:44 Introduction to Nucleophilic Acyl Substitution 08:49 Key Differences from Aldehydes and Ketones: The Role of Leaving Groups 10:01 The General Mechanism for Nucleophilic Acyl Substitution 11:54 The Impact of pH: Acidic vs. Basic Reaction Pathways 15:35 Practical Examples: Basic, Neutral, and Acidic Mechanisms 27:47 Summary and Key Takeaways