Antibiotics as selectable markers (antibiotic-based selection) скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Antibiotics as selectable markers (antibiotic-based selection) в качестве 4k
У нас вы можете посмотреть бесплатно Antibiotics as selectable markers (antibiotic-based selection) или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Antibiotics as selectable markers (antibiotic-based selection) в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Antibiotics as selectable markers (antibiotic-based selection)
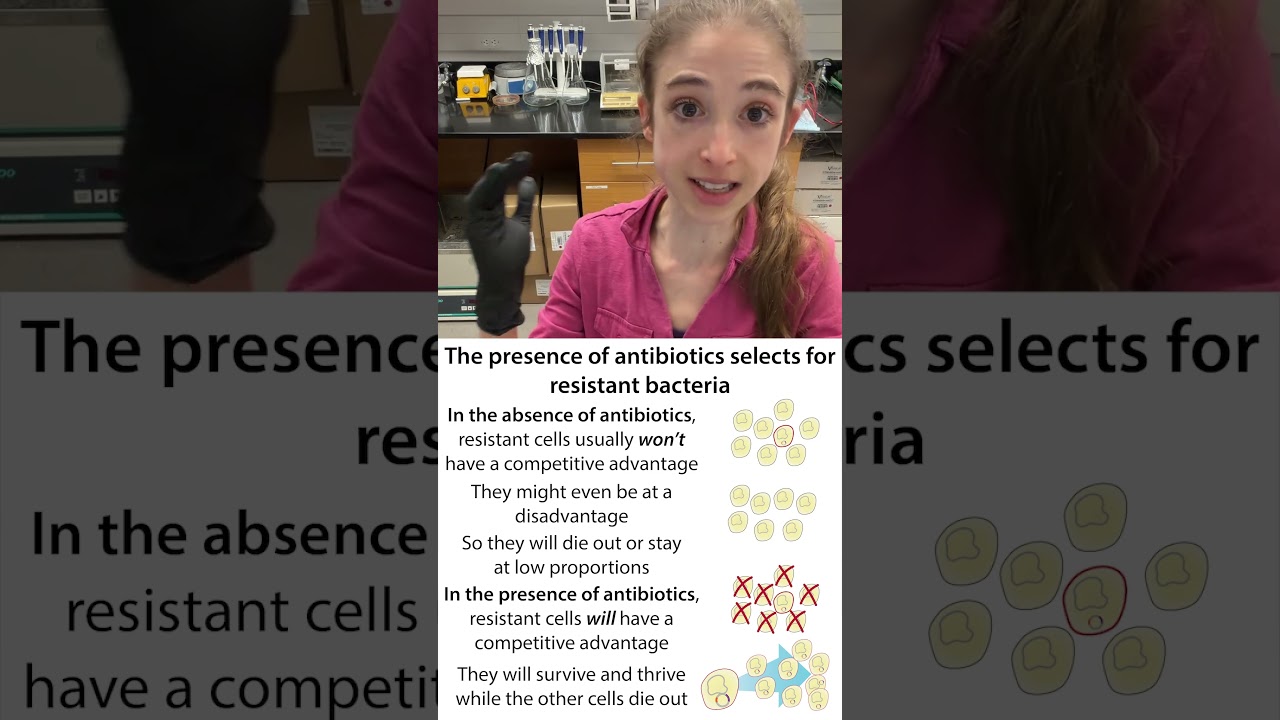
In molecular cloning we stick a gene we’re interested in into a circular piece of DNA called a plasmid then stick that into host cells to make more copies of that gene &/or the protein it codes for. Often those host cells are (harmless) bacteria. They’re single-celled, grow fast, are cheap to feed, & easy to maintain. But sometimes they’re TOO easy to maintain. We only want to take care of the bacteria that are actually doing work for us - they give us DNA & protein & we give them food to pay them back for burdening them & keeping them from making their own stuff. We want to “weed out” any bacteria that aren’t doing our work so we can use antibiotics to select for just ones w/our plasmid. To do this, we put an antibiotic resistance gene into our plasmid alongside our gene, then spike the bacteria food w/the corresponding antibiotic. Before bacteria divide, they replicate their DNA so they can pass down a copy to each daughter cell. Plasmids can replicate a lot and have lots of copies but still, when bacteria divide you can end up w/daughter cells that don’t get a copy. It might seem like no big deal because this doesn’t happen often and, if it does they get killed by the antibiotic you put in to weed out bacteria like them that don’t have the plasmid. BUT those plasmid-less cells don’t have to spend time, energy, & resources making what we want them to make. So, if you take away the antibiotic they have a big growth advantage. So we need to make sure the antibiotics are always present and in tact. To do so: limit growth times Use fresh antibiotic stocks and add them fresh to liquid media right before using Store concentrated (e.g. 1000X) amp aliquots in the freezer and avoid freeze-thaws Don’t add antibiotics to hot media There are some special considerations with ampicillin (Amp) because its antibiotic resistance mechanism is secreting a protein (bectalactamase) to burst the amp outside of cells – which protects surrounding cells. So, for amp selection: Don’t use Amp plates that are more than a few weeks old (or at least use with caution!) Use a higher Amp concentration and/or add more Amp midway through long growths before inoculating a large culture you can spin down cells, remove media, and add fresh media If using a high copy number plasmid, use higher amp concentration (e.g. 100 μg/μL instead of 50 - though I generally just use 100 μg/mL all the time) Consider using a more stable beta-lactam antibiotic like carbenicillin, which is less susceptible to that generic degradation, but more expensive for more on how we take advantage of antibiotic resistance mechanisms in the lab to select for bacteria containing plasmids of interest: https://bit.ly/antibioticselections & • Antibiotic selection - mechanisms & usage ... More on satellite colonies: blog: https://bit.ly/satellitecolonies ; YouTube: • Satellite colonies and ampicillin/beta-lac... and here’s more about ampicillin instability (and how it’s worse that way than other antibiotics): Ryan, K. J., Needham, G. M., Dunsmoor, C. L., & Sherris, J. C. (1970). Stability of antibiotics and chemotherapeutics in agar plates. Applied microbiology, 20(3), 447–451. https://doi.org/10.1128/am.20.3.447-4... more about all sorts of things: #365DaysOfScience All (with topics listed) 👉 http://bit.ly/2OllAB0 or search blog: http://thebumblingbiochemist.com #scicomm #biochemistry #molecularbiology #biology #sciencelife #science #realtimechem