Lipids (Part 2 of 11) - Free Fatty Acids - Intro and Properties скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Lipids (Part 2 of 11) - Free Fatty Acids - Intro and Properties в качестве 4k
У нас вы можете посмотреть бесплатно Lipids (Part 2 of 11) - Free Fatty Acids - Intro and Properties или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Lipids (Part 2 of 11) - Free Fatty Acids - Intro and Properties в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Lipids (Part 2 of 11) - Free Fatty Acids - Intro and Properties
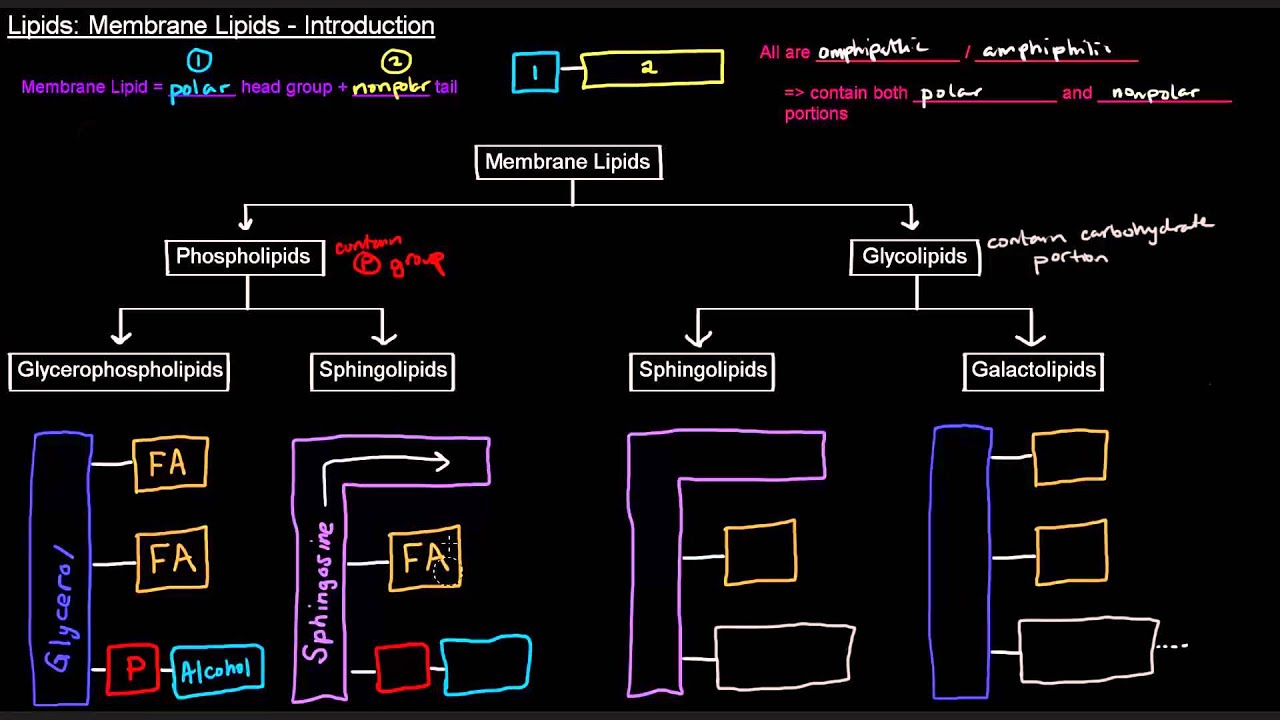
Moof's Medical Biochemistry Video Course: http://moof-university.thinkific.com/... Questions Answered in This Video: What are free fatty acids? What is(are) the function(s) of fatty acids? Why can fatty acids be used for fuel? What is the general structure of a fatty acid? How are fatty acids classified or categorized? What is(are) the difference(s) between a saturated and unsaturated fatty acid? What are cis- and trans- fatty acids? How do chain length and degree of unsaturation relate to melting points of fats? Don't forget to LIKE, COMMENT, and SUBSCRIBE: http://www.youtube.com/subscription_c... INFORMATION ABOUT TUTORING: http://www.moofuniversity.com/tutoring/ TO SUPPORT MOOF UNIVERSITY WITH A FINANCIAL CONTRIBUTION: http://www.moofuniversity.com/support... INSTAGRAM: / moofuniversity FACEBOOK: / 1554858934727545 TWITTER: / moofuniversity Video Content Summary: Free fatty acids are hydrocarbon chains with a carboxylic acid head group and are amphipathic / amphiphilic because of this. They are highly-reduced molecules (more highly reduced than carbohydrates), rich in electrons, and are thus used as fuel. They also function as an important building block for various membrane lipids. At biological pH levels, the carboxylic acid group exists predominantly in its deprotonated carboxylate form. Most often, fatty acids contain an even number of carbon atoms, usually between 4 and 36. Fatty acids are initially classified by the presence or absence of carbon-carbon double bonds in their hydrocarbon chain. Fatty acids that contain zero carbon-carbon double bonds are known as saturated fatty acids - as they are "saturated" with hydrogens. Fatty acids that contain at least one carbon-carbon double bond are known as unsaturated fatty acids. Unsaturated fatty acids can be further classified based on 1) the geometry of the double bond(s) contained within the hydrocarbon chain and/or 2) the number of carbon-carbon double bonds in the hydrocarbon chain. If the carbon-carbon double bond(s) is (are) cis, we have a cis-unsaturated fatty acid, otherwise known simply as a cis fatty acid. If the carbon-carbon double bond(s) is (are) trans, we have a trans-unsaturated fatty acid, otherwise known simply as a trans fatty acid. Further, fatty acids are monounsaturated fatty acids (MUFAs), if they have only one carbon-carbon double bond in the hydrocarbon chain, and polyunsaturated fatty acids (PUFAs), if they have two or more carbon-carbon double bonds in the hydrocarbon chain. Two factors relating to the structures of fatty acids are important to consider when thinking about the melting point of fats: 1) fatty acid chain length and 2) degree of unsaturation / number of cis carbon-carbon double bonds. An increase in chain length increases the number of atoms that can form temporary dipoles, which increases Van der Waals interactions, and thus, increases melting point. A increase in the degree of unsaturation, specifically, an increase in the number of cis double bonds in a fatty acid, will decrease the ability of atoms to form temporary dipoles and thus, decreases Van der Waals interactions and thus, decreases melting point. This is caused by kinks or bends that form in the hydrocarbon chain of a fatty acid due to a cis carbon-carbon double bond. Kinks or bends to not form with trans fatty acids, and they tend to keep a fairly straight chain, similar in structure to that of a saturated fatty acid.