FDA Generic Drugs Forum 2025 - Day 1, Session 3 Q&A скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: FDA Generic Drugs Forum 2025 - Day 1, Session 3 Q&A в качестве 4k
У нас вы можете посмотреть бесплатно FDA Generic Drugs Forum 2025 - Day 1, Session 3 Q&A или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон FDA Generic Drugs Forum 2025 - Day 1, Session 3 Q&A в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
FDA Generic Drugs Forum 2025 - Day 1, Session 3 Q&A
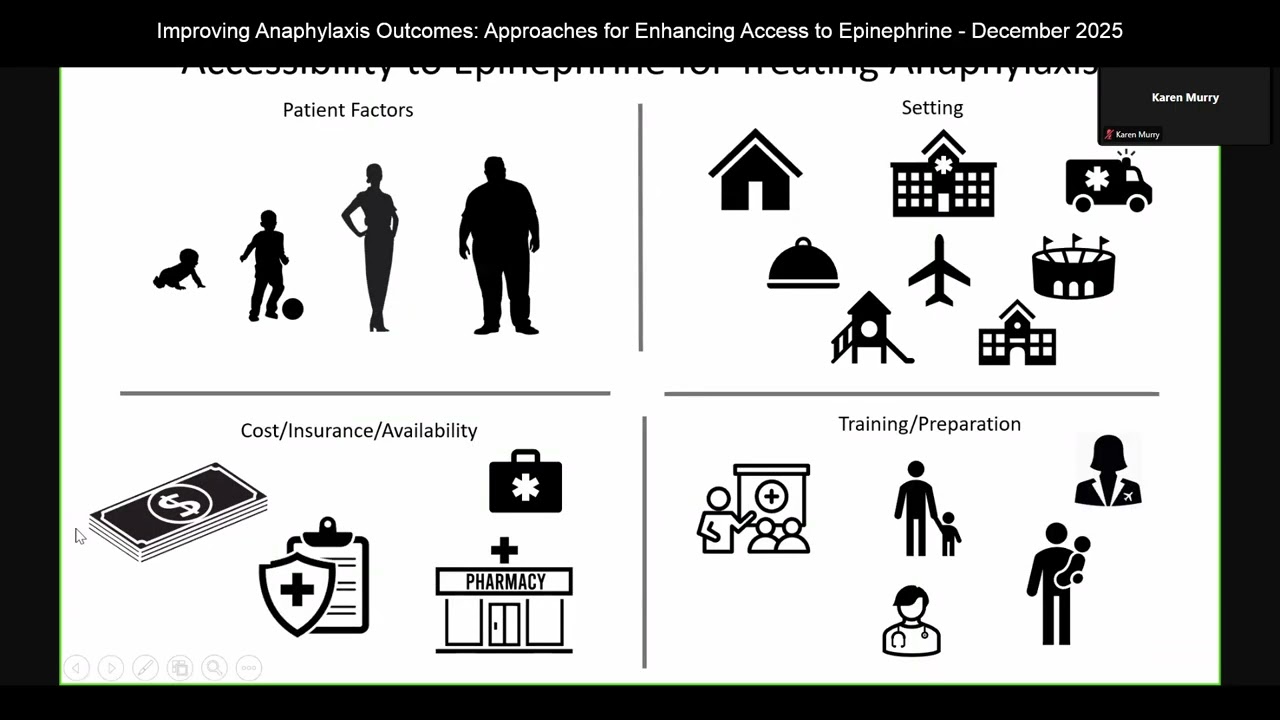
SUBSCRIBE to @FDALearningCache to see more videos. Details and supporting materials: https://fdalearn.com/GDF2025 During the 2025 Generic Drugs Forum, the panel discussed various topics related to excipients in drug formulations, focusing on their safety, evaluation, and regulatory pathways. Questions addressed the use of new excipients in generics, with experts noting that while new excipients require safety data, their inclusion is typically evaluated through the NDA pathway. There were also discussions on excipient interactions with active pharmaceutical ingredients (APIs), such as the historical example of tetracycline and citrate, with panelists clarifying that only known interactions are actively assessed. Other topics included the importance of proper excipient identification through UNI codes for consistency in evaluations and the potential for harmonizing international guidelines for excipient use. Panelists also provided insights into the challenges in evaluating excipients for topical drug products and the need for safety justification when proposing different excipients compared to reference products. The session concluded with an acknowledgment of the limitations of the IID database, such as its lack of context of use information, and a brief mention of ongoing discussions about updating the database.