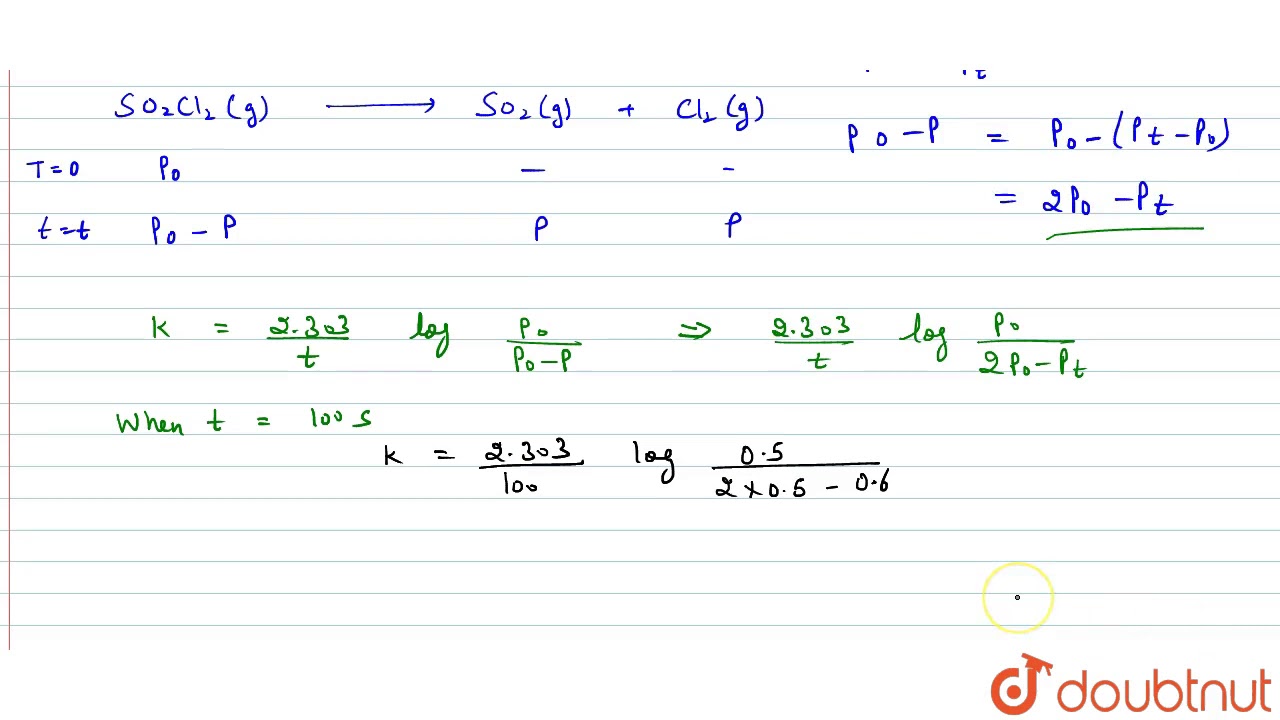The following data were obtained during the first order thermal decomposition of SO₂Cl₂ at constant скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: The following data were obtained during the first order thermal decomposition of SO₂Cl₂ at constant в качестве 4k
У нас вы можете посмотреть бесплатно The following data were obtained during the first order thermal decomposition of SO₂Cl₂ at constant или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон The following data were obtained during the first order thermal decomposition of SO₂Cl₂ at constant в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
The following data were obtained during the first order thermal decomposition of SO₂Cl₂ at constant
Unlock the secrets of Chemical Kinetics with detailed NCERT-based solutions for Class 12 Chemistry. In this video, Chemistry expert OSB breaks down key concepts, solved examples, and important tricks for CBSE Board, NEET, and JEE aspirants. Perfect for last-minute revision and conceptual clarity! The following data were obtained during the first order thermal decomposition of SO₂Cl₂ at constant volume. Reaction: SO₂Cl₂(g) → SO₂(g) + Cl₂(g) Calculate the rate of the reaction when total pressure is 0.65 atm. 📘 Topics Covered: • Rate of Reaction • Integrated Rate Law • First & Zero Order Reactions • Units of Rate Constant • Arrhenius Equation • Graphical Tricks & Mistake Traps 🔍 Ideal for Class 12 CBSE students, NEET/JEE aspirants, and anyone aiming for high scores with smart strategies. 🎯 Explained by: OSB – CBSE Chemistry Expert Subscribe for more NCERT solutions, exam tips, and powerful shortcuts! ⸻ 20 Hashtags: #ChemicalKinetics, #Class12Chemistry, #NCERTSolutions, #CBSEChemistry, #NEET2025, #JEE2025, #ChemistryWithOSB, #CBSEBoard2025, #ScienceWithOSB, #RateOfReaction, #ArrheniusEquation, #ChemistryTricks, #IntegratedRateLaw, #CBSETipsAndTricks, #ZeroOrderReaction, #FirstOrderReaction, #JEEChemistry, #NEETChemistry, #ChemistryRevision, #NcertClass12


![For the reaction: the rate = k[A][B]2 with k = 2.0 × 10–6 mol–2 L2 s–1. Calculate the initial rate..](https://imager.clipsaver.ru/tu2tl37gQ_c/max.jpg)