Basics of Clinical Research 1 скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Basics of Clinical Research 1 в качестве 4k
У нас вы можете посмотреть бесплатно Basics of Clinical Research 1 или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Basics of Clinical Research 1 в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Basics of Clinical Research 1
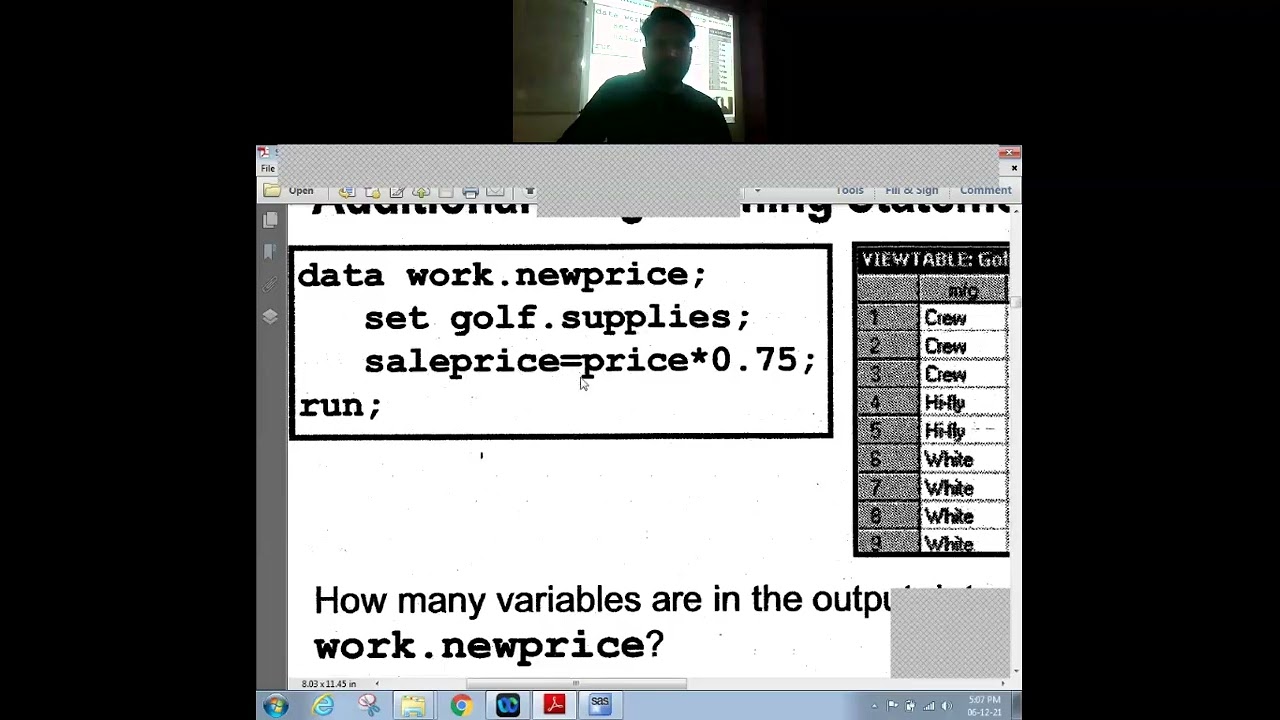
Introduction to Clinical Researc What is Drug? Types of Drug Routes of Drug Administration Drug Approval History Jack-Leg Paralysis (1930) Elixir Sulfanilamide-Taste of Death (1937) Thalidomide Tragedy (1962) : 50% death from total case Need for Drug Regulation Functions of CDSCO Drug Application (IND, NDA, ANDA) Drug Development History Drug Approval Process in India Drug Discovery Trials Drug Development Process Flow What is Clinical Trials? Types of Clinical Trials Drug Discovery in Lab Testing Choosing a Disease and Find a Drug Decide Lead Compound Lead Identification Molecule Target Identification & Validation Pre-Clinical in Animal Testing Trials: In VIVO & VITRO Trials Toxicology Trials Pharmacology Trials Haematology Parameters Biochemistry Parameters MTD LD50 Type of Clinical Trials: (Find Dose Ranging, Tolerability, Safety, PK and Efficacy) Phase I Clinical Trials Phase II Clinical Trials (SAD study, MAD study) Phase III Clinical Trials Phase IV Clinical Trials (Post Market Surveillance) Study Design / Trials: Equivalence Design Superior Design Non-Interior Design Experimental Design Observational Design Randomized Design Non-Randomized Design Type of Study / Design: Cohort Study Case-Control Study Cross-Sectional Study Prospective / Longitudinal / Forward Study Retrospective / Backward Study Snap Shot Study Crossover Study Parallel Design Replicate Design for Highly Variable Drug Partial Replicate Design Fully Replicate Design Some Important Topic in Clinical Research: Half Life Washout Period Carry over Effect Intra Subject Variability Inter Subject Variability Bioavailability & Bio-equivalence Trials Pharmacokinetic and Pharmacodynamics Trials Drug Movement in the body Pharmacokinetic Parameters Cmax, AUCo-t, AUCo-inf, tmax, t1/2,etc. PK Concentration Data PK Derived Parameters Data Steady State Study Introduction to Clinical Research: Protocol Synopsis ICF ( Informed Consent Form) CRF (Case Report Form) SOPs in Clinical trials SAP (Statistical Analysis Plan) Subject vs Patient Efficacy And Safety Placebo Effect Open Label Double Blind Trials Inclusion/Exclusion Criteria Ethical guidelines in Clinical Research: GCP (Good Clinical Practice) GMP (Good Manufacturing Practice) ICH (The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use) ICHE3 ICH E6 (Good Clinical Practice) ICH E9 (Statistical Principal for Clinical trials) IEC (Independent Ethics Committee ) IRB (Institutional Review Board) Regulatory guidelines in Clinical Research: IND ( Investigational New Drug) ANDA(Abbreviated New Drug Application) NDA (New Drug Application) Regulatory in Clinical Research: DCGI USFDA MHRA ANVIZA CANADA,etc Different Departments and Work Profiles in Clinical Research Organizations: Clinical Departments Analytical Department Clinical Data Management Department Biostatistics & Programming Department Quality Control Department Quality Assurance Department Regulatory Department Medical Writing Department Pharmacovigilance Department Project Management Department Business Development Department Roles and Responsibilities in Clinical Research: Sponsor Investigator IEC (Independence Ethics Committee ) IRB (Institutional Review Board) DMC (Data Monitoring Committee) CRA (Clinical Research Associate) CRC (Clinical Research co-ordinator) Auditors Data Manager Statistician SAS Programmer QA(Quality Assurance) QC (Quality Control) Some Important topics in Clinical Research: Feasibility Subject Recruitment Procedure Screening and Sample collection Dosing and Storage Washout Period Calculation Framing of PK Time Points Interim Analysis Primary Endpoint Secondary Endpoint Download the SaSTAT app today and start your journey towards mastering SAS! 🌎 https://eduthanos.page.link/qY6q (Org Code: UULCGK) Get in touch at: 95373 77574 Website:- www.sastat.com #SaSTAT #Ahmedabad #Gujarat #India #Datascience #ClinicalSASInternship #SASCourse #SASTraining #SASProgramming #BaseSASCourse #ClinicalSASTraining #ClinicalSASProgrammer #SASProgrammerJob #SASJobInIndia #SASCourseInIndia #SASTrainingInGujarat #SASCourseForFreshers #CareerInSAS #SASRoadmap #CompleteSASRoadmap #HowToBecomeSASProgrammer #HowToGetSASJob #ClinicalSASJobForFreshers #SASInterviewQuestions #DataAnalyticsCourse #DataAnalystCareer #PharmaDataAnalytics #WhyClinicalSASPaysHigh #OnlineSASCourse #LearnSASFrom #SASCertification