Oxygen vs Water | Why One Is Stable & One Isn't скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Oxygen vs Water | Why One Is Stable & One Isn't в качестве 4k
У нас вы можете посмотреть бесплатно Oxygen vs Water | Why One Is Stable & One Isn't или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Oxygen vs Water | Why One Is Stable & One Isn't в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Oxygen vs Water | Why One Is Stable & One Isn't
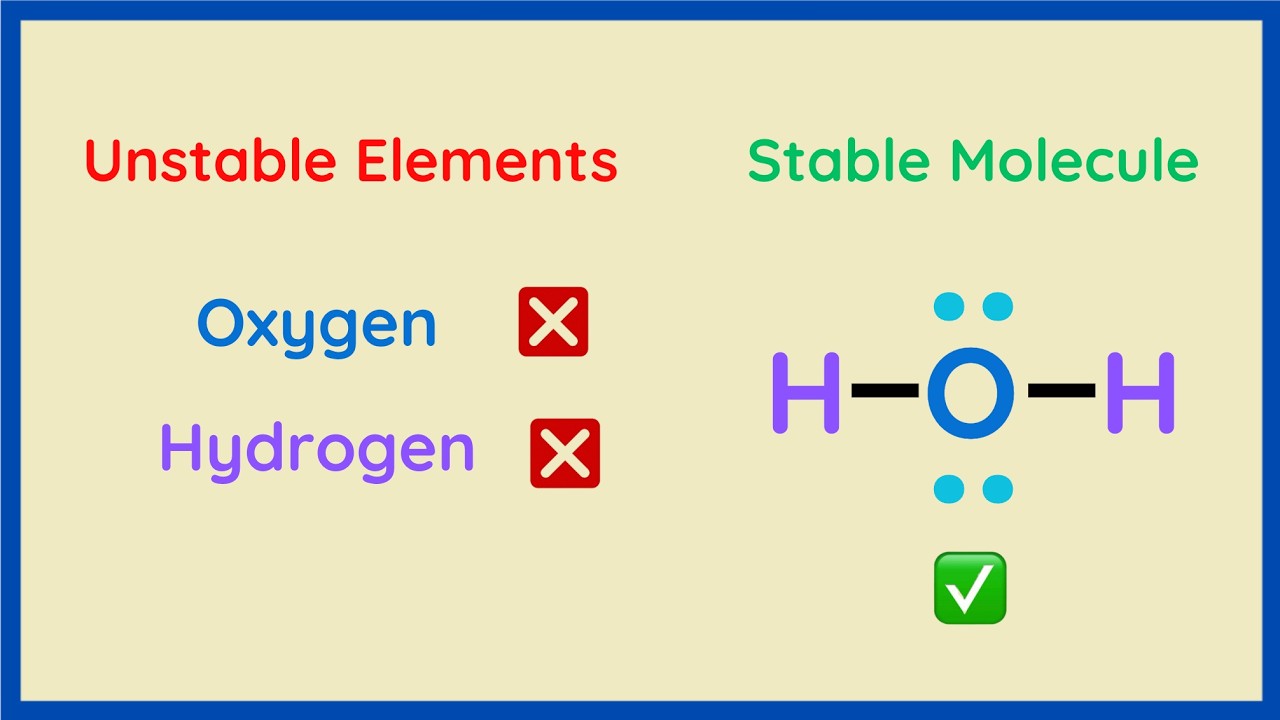
Discover the fundamental principles of molecular stability by examining water (H2O) as a prime example. We illustrate how individual unstable elements form stable bonds to achieve fully-filled electron shells, a core concept in chemistry. This video clarifies the role of electron valence and valence electrons in determining stability, connecting these ideas to the atomic structure and the periodic table in a way that is easily chemistry explained. Is water (H₂O) a stable molecule or unstable? 🤔 In this video, we break down why water, made from the “unstable” elements hydrogen and oxygen, ends up being so stable! We explore: The composition of water using Lewis structures The difference between dots and sticks in electron bonding How valence electrons determine atomic stability The octet rule and why some elements like Neon are naturally stable How hydrogen and oxygen achieve stability through bonding If you’ve ever wondered why H₂O molecules don’t fall apart even though they’re made of reactive elements, this video explains it in clear, step-by-step visual terms. Perfect for chemistry students, enthusiasts, or anyone curious about the science behind water! 💧 Watch to discover: ✅ Why H and O are unstable alone but stable in H₂O ✅ How bonds share electrons to satisfy the octet (or duet!) rule ✅ How the periodic table predicts stability and reactivity #Chemistry #WaterMolecule #LewisStructure #OctetRule #ValenceElectrons #H2O #MoleculeStability #ScienceExplained



















