ClO4- Lewis Structure - How to Draw the Lewis Structure for ClO4- (Perchlorate Ion) скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: ClO4- Lewis Structure - How to Draw the Lewis Structure for ClO4- (Perchlorate Ion) в качестве 4k
У нас вы можете посмотреть бесплатно ClO4- Lewis Structure - How to Draw the Lewis Structure for ClO4- (Perchlorate Ion) или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон ClO4- Lewis Structure - How to Draw the Lewis Structure for ClO4- (Perchlorate Ion) в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
ClO4- Lewis Structure - How to Draw the Lewis Structure for ClO4- (Perchlorate Ion)
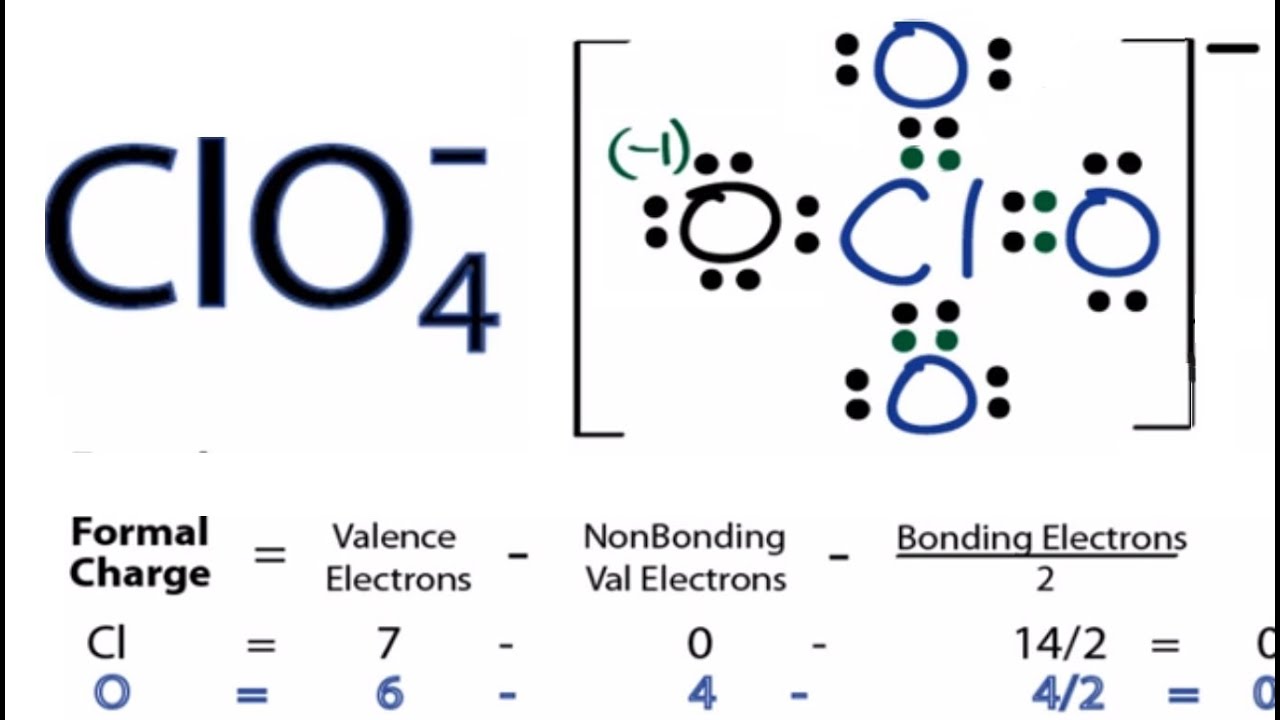
A step-by-step explanation of how to draw the ClO4- Lewis Structure (Perchlorate Ion). The ClO4-Lewis structure is a good structure to help you understand why calculating formal charges is important. The first structure you come up with might work in terms of filling the octets of each atom but you should check the formal charges for ClO4-since Chlorine (Cl) is in Period Three on the Periodic Table and can hold more than eight valence electrons. For the ClO4- Lewis structure the total number of valence electrons (found on the periodic table) for the ClO4-molecule. Once we know how many valence electrons there are in ClO4- we can distribute them around the central atom with the goal of filling the outer shells of each atom. Only use the number of available valence electrons we calculated earlier. Also note that you should put the ClO4- Lewis structure in brackets with a negative sign on the outside to show that it is an ion with a negative one charge. Note that ClO4- can have an Expanded Octet and have more than eight valence electrons. Because of this there may be several possible Lewis Structures. To arrive at the most favorable Lewis Structure we need to consider formal charges. See how to calculate formal charges: • Formal Charges: Calculating Formal Charge ---- Steps to Write Lewis Structure for compounds like ClO4 ------ 1. Find the total valence electrons for the ClO4- molecule. 2. Put the least electronegative atom in the center. Note: Hydrogen (H) always goes outside. 3. Put two electrons between atoms to form a chemical bond. 4. Complete octets on outside atoms. 5. If central atom does not have an octet, move electrons from outer atoms to form double or triple bonds. ---- Lewis Resources ---- • Lewis Structures Made Simple: • How to Draw Lewis Structures: Five Easy Steps • More practice: • Lewis Dot Structure Practice Problems (wit... • Counting Valence Electrons: • Finding the Number of Valence Electrons fo... • Calculating Formal Charge: • Formal Charges: Calculating Formal Charge • Exceptions to the Octet Rule: • Exceptions to the Octet Rule Lewis Structures are important to learn because they help us understand how atoms and electrons are arranged in a molecule, such as Chlorate ion. This can help us determine the molecular geometry, how the molecule might react with other molecules, and some of the physical properties of the molecule (like boiling point and surface tension). Chemistry help at https://www.Breslyn.org

















![Эффект Джанибекова [Veritasium]](https://imager.clipsaver.ru/N9HlQ-XVnFk/max.jpg)

