iMeta | Antioxidants promote metabolic remodeling in rumen epithelium through single-cell resolution скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: iMeta | Antioxidants promote metabolic remodeling in rumen epithelium through single-cell resolution в качестве 4k
У нас вы можете посмотреть бесплатно iMeta | Antioxidants promote metabolic remodeling in rumen epithelium through single-cell resolution или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон iMeta | Antioxidants promote metabolic remodeling in rumen epithelium through single-cell resolution в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
iMeta | Antioxidants promote metabolic remodeling in rumen epithelium through single-cell resolution
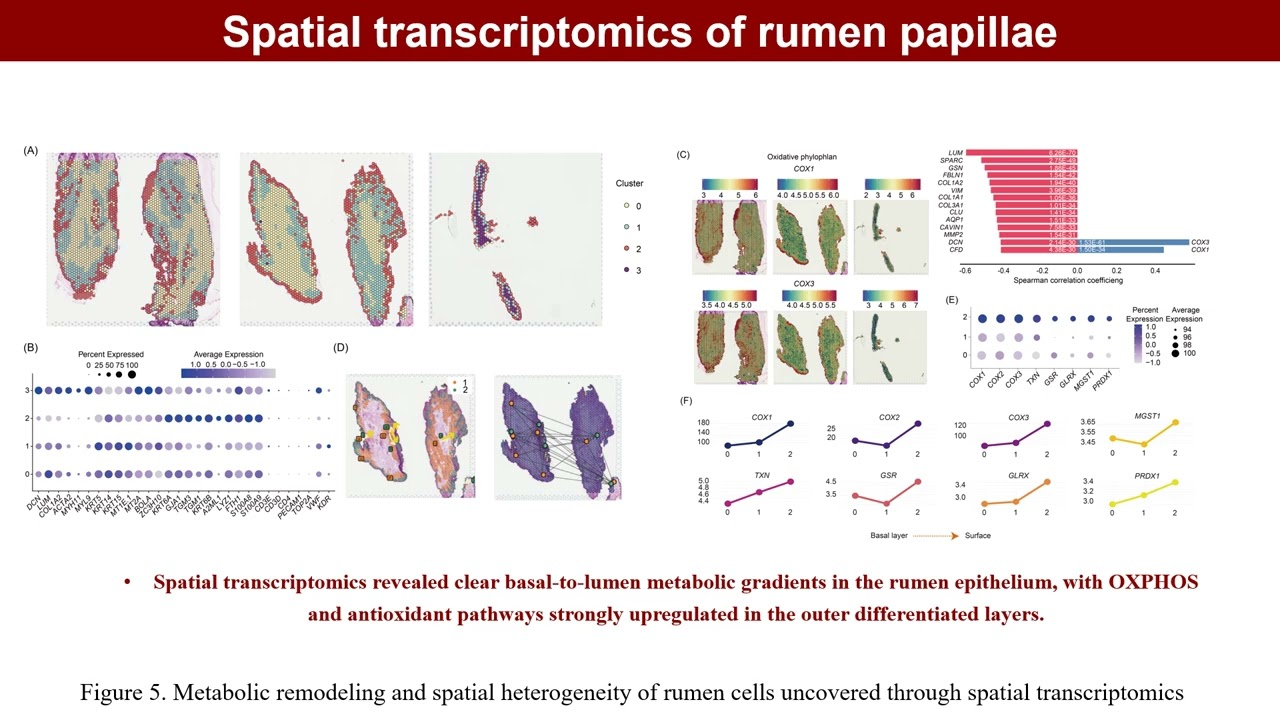
RESEARCH ARTICLE Open Access Antioxidants promote metabolic remodeling in cattle rumen epithelium revealed by single-cell resolution Sen-Lin Zhu, Yu-Nan Yan, Ming-Hui Jia, Hou-Cheng Li, Bo Han, Tao Shi, Lian-Bin Xu, Xiao-Wen Wang, Qi Zhang, Wei-Jie Zheng, Jing-Hong Xu, Liang Chen, Wenlingli Qi, Sheng-Jun Cai, Xin-Peng Chen, Feng-Fei Gu, Jian-Xin Liu, George E. Liu, Yu Jiang, Dong-Xiao Sun, Ling-Zhao Fang, Hui-Zeng Sun (Yu-Nan Yan, Ming-Hui Jia, Hou-Cheng Li, Bo Han, Tao Shi, Lian-Bin Xu, and Xiao-Wen Wang contributed equally to this study.) Abstract Oxygen signaling is essential for cellular homeostasis and tightly linked to metabolism, growth, and survival. In dairy cows, oxidative stress, arising from an imbalance between reactive oxygen species and antioxidants, is a major postpartum challenge that contributes to disease susceptibility. Using single-cell transcriptomes from 1,793,854 cells across 59 tissues, we analyzed oxygen signaling states within 1006 cellular clusters. The gastrointestinal tract (GIT) epithelium, particularly the forestomach, exhibits the strongest antioxidant activity, closely coupled to oxidative phosphorylation (OXPHOS) and glycolysis, with OXPHOS levels surpassing those of cardiomyocytes and hepatocytes (Cohen's d> 3.9, p <0.001). Pseudotime and spatial transcriptomics demonstrated that both OXPHOS and antioxidant capacity increase progressively along the basal-to-luminal differentiation axis. Functional experiments in primary rumen epithelial cells showed that antioxidant supplementation or GPX1 modulation enhances mitochondrial respiration, boosts intracellular glutathione, and accelerates epithelial differentiation. Limited proteolysis-mass spectrometry (LiP-MS) analysis identified GPX1, GSTP1, COX7A2, and COX6B1 as candidate targets mediating antioxidant-driven metabolic remodeling. Together, these results reveal a redox-governed metabolic program in the forestomach epithelium and highlight antioxidant interventions as a potential strategy to support epithelial development and mitigate oxidative stress-related disorders in dairy cattle. Highlights A single-cell transcriptomic atlas of 1.79 million bovine cells revealed the forestomach epithelium as one of the most antioxidant-active tissues. As the forestomach epithelium differentiates from the basal layer towards the luminal surface, both mitochondrial respiration and antioxidant capacity progressively intensify, establishing a coupled metabolic–redox gradient. Functional assays identified GPX1 as a key antioxidant target mediating OXPHOS and cell differentiation.



















