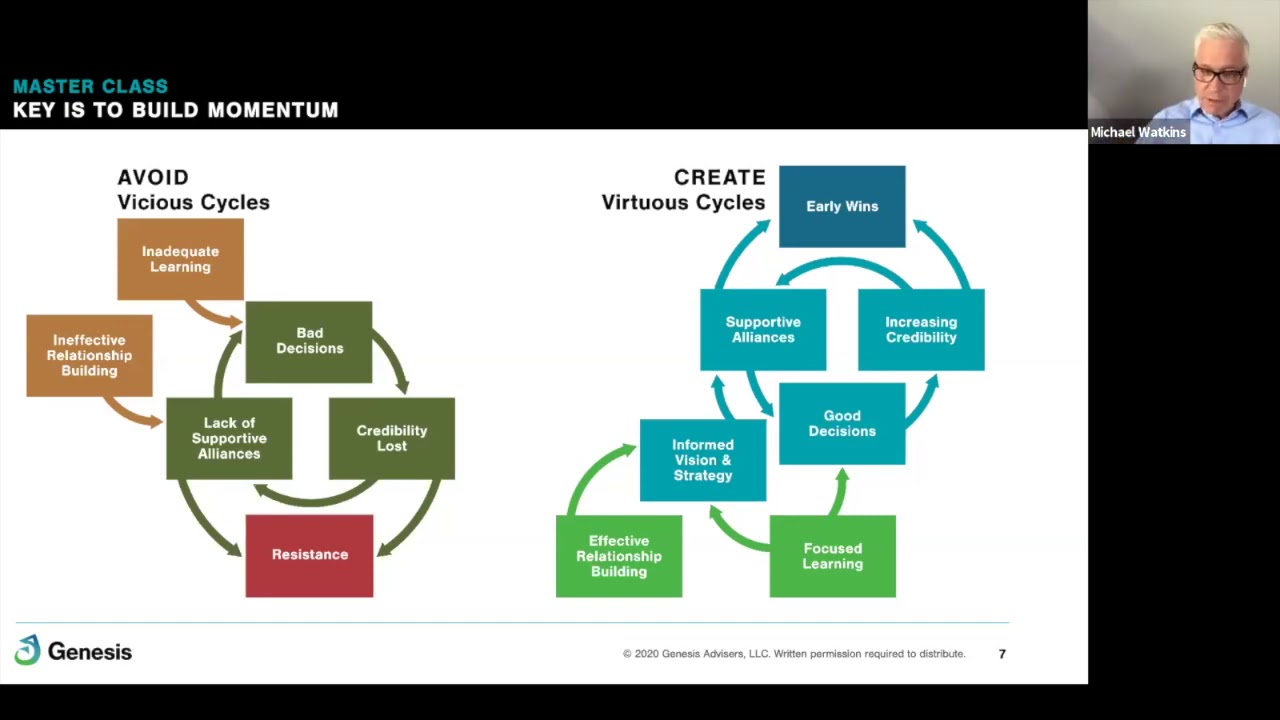
GCC RA Pharma Summit 2025 - Day 2 | Session 2: Strengthening Global Health Systems скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: GCC RA Pharma Summit 2025 - Day 2 | Session 2: Strengthening Global Health Systems в качестве 4k
У нас вы можете посмотреть бесплатно GCC RA Pharma Summit 2025 - Day 2 | Session 2: Strengthening Global Health Systems или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон GCC RA Pharma Summit 2025 - Day 2 | Session 2: Strengthening Global Health Systems в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
GCC RA Pharma Summit 2025 - Day 2 | Session 2: Strengthening Global Health Systems
-18th-Feb-2025 Moderated by: Dr. Jacqueline Acquah Senior Regulatory Affairs Lead, Middle East & Africa Coalition for Epidemic Preparedness Innovations (CEPI) Keynote presentation, by: Advancing Global Health: CEPI’s Role in Regulatory Preparedness and Vaccine R&D Innovation Dr. Alessandro Lazdins Regulatory Policy and Intelligence Manager CEPI Dr. Lazdins presented CEPI’s role in accelerating vaccine development and strengthening regulatory systems to ensure timely responses to emerging health threats. Panel discussion, featuring: The session continued with a panel of distinguished experts from across the GCC and wider region, who shared experiences on regulatory frameworks, vaccine evaluation, and preparedness: Dr. Anees Puthan Veettil – Head of Evaluation Unit of Pharmaceutical Studies, Drug Department, Emirates Drug Establishment (EDE), UAE Dr. Abeer Althiban – Scientific Evaluation Expert, Infectious and Immunity Section, Saudi Food & Drug Authority (SFDA) Dr. Rawan I. Aloufi – Laboratory Expert, Vaccines Lab Department, Biologics Executive Department, Saudi Food & Drug Authority (SFDA) Dr. Reem Mahmoud Eltanahy – Head of Variation Unit for Biological Products, Egyptian Drug Authority (EDA) For more information, please visit: https://pramagcc.com