What is stress corrosion? Stress corrosion cracking/Stress cracking: Electrochemical Corrosion скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: What is stress corrosion? Stress corrosion cracking/Stress cracking: Electrochemical Corrosion в качестве 4k
У нас вы можете посмотреть бесплатно What is stress corrosion? Stress corrosion cracking/Stress cracking: Electrochemical Corrosion или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон What is stress corrosion? Stress corrosion cracking/Stress cracking: Electrochemical Corrosion в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
What is stress corrosion? Stress corrosion cracking/Stress cracking: Electrochemical Corrosion
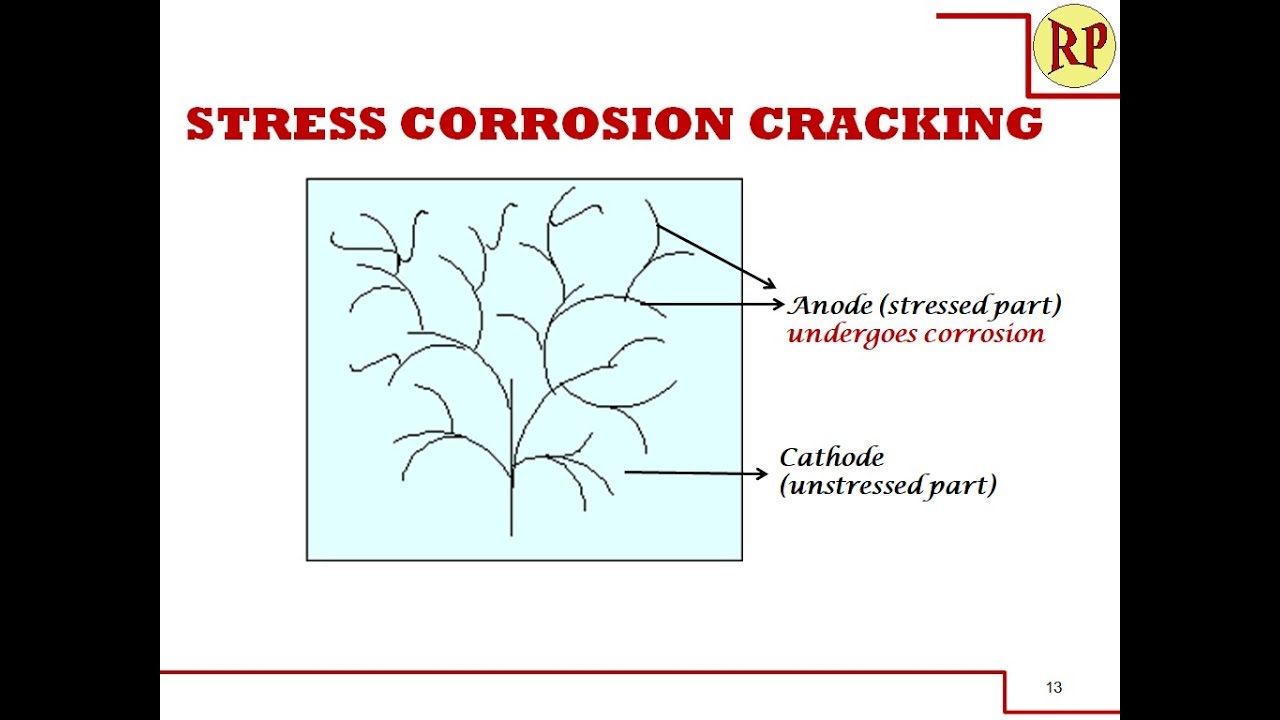
This explains the stress corrosion and the conditions. Three different mechanisms involved in stress corrosion. Explains caustic embrittlement, season cracking, hydrogen embrittlement. Preventive measures of stress corrosion. Stress Corrosion/Stress Cracking/Stress Corrosion Cracking You will be able to define stress corrosion. You will be able to enumerate the conditions and causes for stress corrosion. You will be able to explain the mechanism of stress corrosion with special cases . You will be able to suggest the preventive measures for stress corrosion. Stress corrosion Stress corrosion occurs due to the tensile stresses (applied stress and residual stress) on the susceptible material in the presence of a specific corrosive environment, resulting in the formation of crack which propagates. Causes of Stress corrosion Susceptible metal Specific corrosive environment Copper alloys NH3 and ammonia compounds Mild steel Alkalis and nitrates Stainless steel Chlorides and acid chlorides Causes of Stress corrosion Due to stresses caused by heavy machining works in fabricated articles of certain alloys Rolling Drawing Insufficient annealing Cold or hot bending Welding Mechanism Embrittlement Caustic embrittlement Hydrogen embrittlement. Active Path Dissolution Film induced cleavage Caustic embrittlement in high pressure steam boilers Carbon and low alloy steels – in the presence of caustic solutions, nitrates, phosphates, carbonates Na2CO3(used for water softening) present in boiler feed water decomposes NaOH flows into hair cracks of boiler walls by capillary action and reacts with iron to form sodium ferroate Season cracking in copper alloys Due to high residual stresses caused by cold working in fabricated articles of brass in the presence of ammonia or amines. Dissolution of brass in ammonia leads to intergranular corrosion Hydrogen embrittlement Hydrogen being small, dissolves in all metals and diffuse between metal atoms in the crystal lattice. Active Path Dissolution Eg. Precipitation of chromium carbide over stainless steel. Chromium concentration is low along the grain boundaries (susceptible region) – less passivated (anode) - corrosion occurs Unstressed region – more passivated (cathode) Film induced cleavage Brittle film over ductile metal-Crack induced in the film and propagates into ductile metal -If brittle film is formed due to corrosion process, it repeats till transgranular fracture occurs Prevention of stress corrosion Avoid specific chemical environment. Control operating temperature. Control stresses while machining. Choose materials which do not react with the specific chemical environment.



















