Targeting Hippo Pathway Cancers: Insilico's ISM6331 (Pan-TEAD) Doses First Patient скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Targeting Hippo Pathway Cancers: Insilico's ISM6331 (Pan-TEAD) Doses First Patient в качестве 4k
У нас вы можете посмотреть бесплатно Targeting Hippo Pathway Cancers: Insilico's ISM6331 (Pan-TEAD) Doses First Patient или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Targeting Hippo Pathway Cancers: Insilico's ISM6331 (Pan-TEAD) Doses First Patient в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Targeting Hippo Pathway Cancers: Insilico's ISM6331 (Pan-TEAD) Doses First Patient
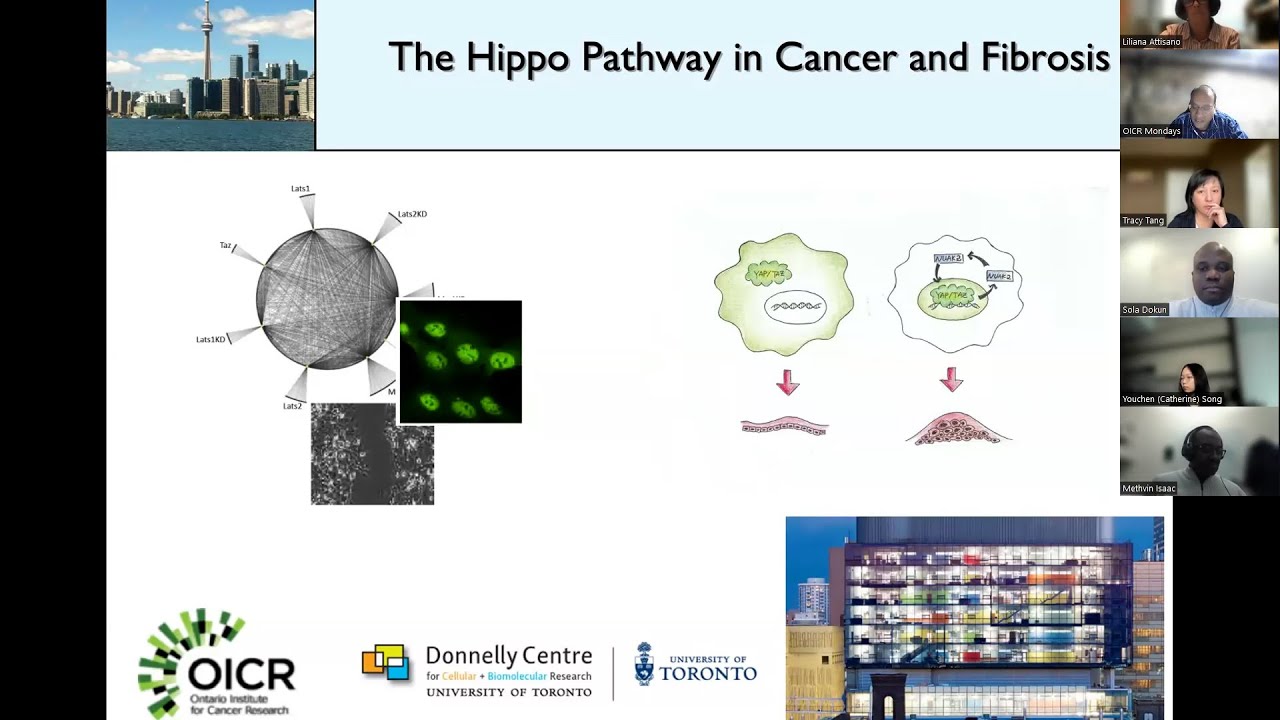
The Hippo pathway is a critical biological signaling system that controls organ size and suppresses tumor growth by regulating cell division and death. When mutated or dysregulated, this pathway becomes a devastating engine of cancer progression, and yet no targeted therapy is available. The core failure occurs when the pathway is inactivated, allowing uncontrolled activation of TEAD transcription factors. These proteins bind to co-activators like YAP/TAZ, flooding the nucleus with signals that drive uncontrolled cell proliferation, block cell death, and promote metastasis. Cancers like malignant mesothelioma, epithelioid hemangioendothelioma, and certain glioblastomas are especially dependent on this hijacked biology, leading to aggressive, treatment-resistant tumors with poor survival rates. Insilico Medicine’s ISM6331 attacks this problem at its source as a pan-TEAD inhibitor. Unlike selective inhibitors targeting single TEAD isoforms (TEAD1, 2, 3, or 4), ISM6331 potently inhibits all four. This versatility is critical because tumors exploit multiple TEAD family members to sustain growth and evade therapy. By broadly blocking TEAD’s palmitoylation pocket—the same site where natural co-activators bind—ISM6331 collapses the entire oncogenic transcription complex. Preclinical data confirms its pan-TEAD action disrupts tumor survival signals across diverse Hippo-dysregulated cancers while showing low-nanomolar potency. Developed using Insilico’s generative AI engine Chemistry42, ISM6331’s novel non-covalent structure achieves unprecedented selectivity. It avoids off-target effects while enabling synergistic potential with chemotherapy, immunotherapy, or targeted agents—crucial for overcoming resistance in advanced tumors. In January 2025, the first patient was dosed in a global multi-center Phase I trial across U.S. and Chinese sites.

















![[Nature Communications] Oral ENPP1 Inhibitor as Next Generation STING Modulator for Solid Tumors](https://imager.clipsaver.ru/R9wa1NeqVZg/max.jpg)
