Meaningful Clinical Endpoints with Electroretinography скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Meaningful Clinical Endpoints with Electroretinography в качестве 4k
У нас вы можете посмотреть бесплатно Meaningful Clinical Endpoints with Electroretinography или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Meaningful Clinical Endpoints with Electroretinography в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Meaningful Clinical Endpoints with Electroretinography
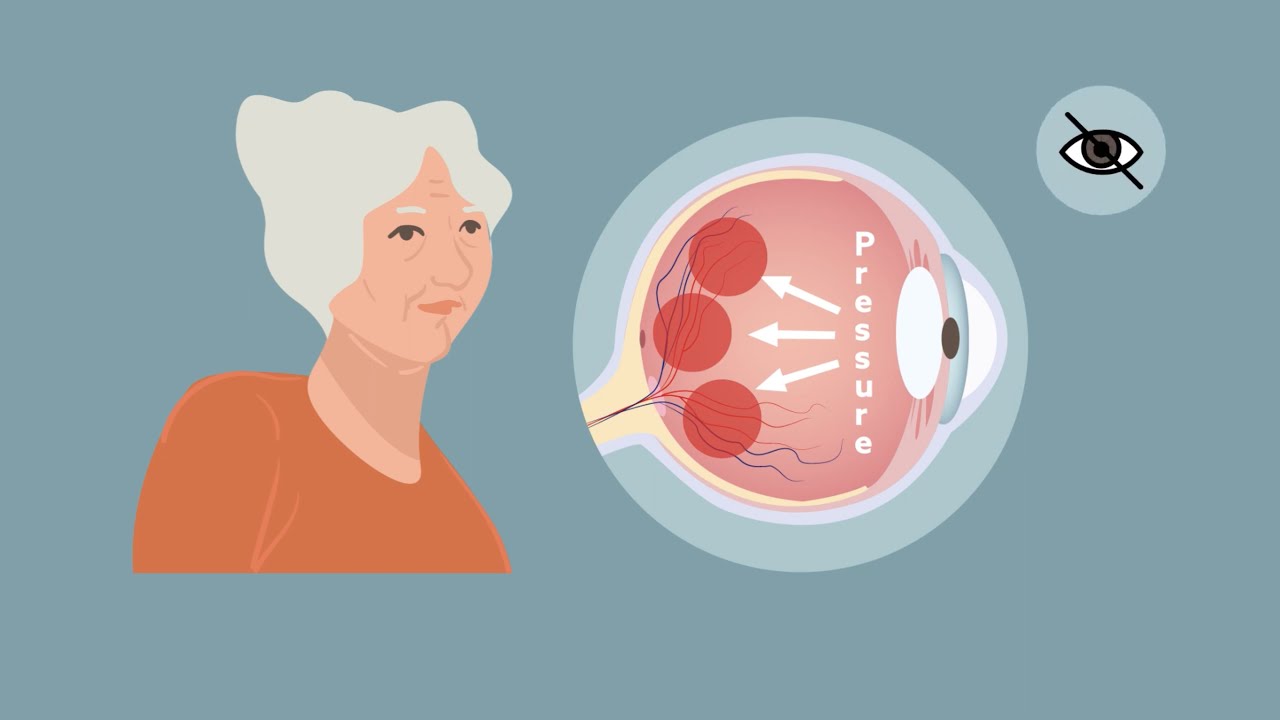
Originally presented at a dinner event at ARVO 2020 entitled: "Meaningful Clinical Endpoints with ERG: Why Detecting Functional Change before Structural Change Matters" The talk centered on the handheld and non-mydriatic RETeval Device for ERG testing that is rapid, non-invasive, and ideal for various endpoint tracking for pharmaceutical or clinical research in the ocular space. Dr. Mitch Brigell, PhD presented on practical electroretinographic testing with the RETeval as both a clinical efficacy and a translational biomarker. It provides, for example, a functional measure of diabetic retinopathy risk progression that was more predictive than structural imaging. Likewise, functional testing of the ganglion cell layer (PhNR) provides an objecting measure of glaucoma. Dr. Quentin Davis, PhD, presented on the scientific and logistical rationale for conducting clinical trials with ERG testing using the RETeval Device. The reliability and relatively inexpensive cost of the unit, combined with it's rapid and non-invasive interface makes it ideal for efficacy endpoint tracking in clinical trials related to ocular disease. Learn more at lkc.com Mitch Brigell, PhD 00:00-Intro 00:40-RETeval: Practical ERG Technology for Clinical Trials 2:51-Prognostic Value of ERG in Diabetic Retinopathy 12:36-Exploratory Endpoints: Phase 2B Study in NPDR patients 18:54-ERG Photopic Negative Response (PhNR) for Ganglion Cell Activity (e.g. Glaucoma) 22:54-Safety: The FDA's Viewpoint on Electroretinography 30:45-Translational ERG Testing in Preclinical Models 33:50-Preclinical ERG Use in Safety: Pharmacology and Toxicology 34:58-Mitch Brigell, PhD: Summary Quentin Davis, PhD 35:44-Scientific Rationale for why the RETeval Device is the essential ERG device for clinical studies 40:58-Logistical rationale for deploying the RETeval Device in clinical studies 41:53-Video demo of the RETeval Device in action 43:51-The RETeval Device ensures quality results 47:21-Post-test quality checks (with examples) 54:01-Quentin Davis, PhD: Summary




![Пожалуй, главное заблуждение об электричестве [Veritasium]](https://imager.clipsaver.ru/6Hv2GLtnf2c/max.jpg)