AS 9701 Organic Chemistry Part 13 | Halogenoalkanes | Nucleophilic Substitution скачать в хорошем качестве
Повторяем попытку...
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: AS 9701 Organic Chemistry Part 13 | Halogenoalkanes | Nucleophilic Substitution в качестве 4k
У нас вы можете посмотреть бесплатно AS 9701 Organic Chemistry Part 13 | Halogenoalkanes | Nucleophilic Substitution или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон AS 9701 Organic Chemistry Part 13 | Halogenoalkanes | Nucleophilic Substitution в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
AS 9701 Organic Chemistry Part 13 | Halogenoalkanes | Nucleophilic Substitution
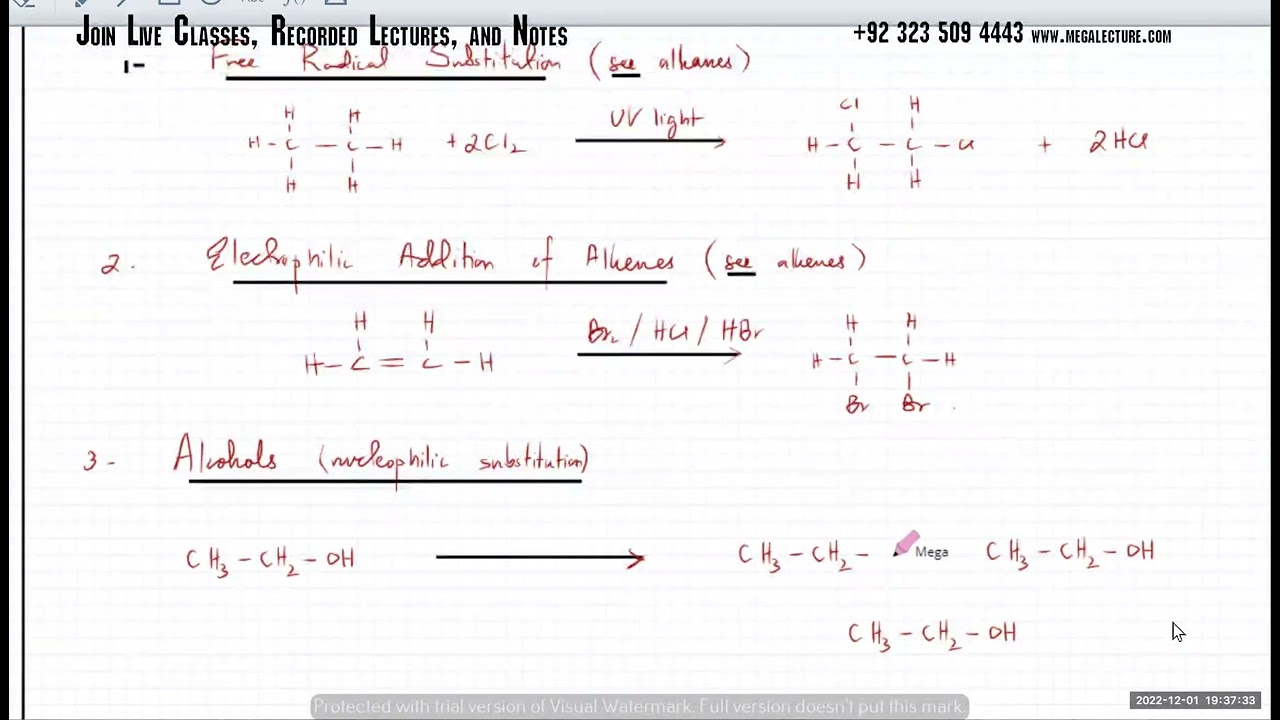
To join our Free Trial Lesson, Join our Whatsapp Community Now! https://chat.whatsapp.com/CWSRaMi4i3w... or msg at +92 323 509 4443 To purchases recorded courses with notes https://megalecture.com/courses/ Book a FREE demo class for Oct Nov 2022 session through the link below: https://docs.google.com/forms/d/e/1FA... Visit our website for video courses and other resources: https://megalecture.com/courses/ 15 Halogen compounds 15.1 Halogenoalkanes Learning outcomes Candidates should be able to: 1 recall the reactions (reagents and conditions) by which halogenoalkanes can be produced: (a) the free-radical substitution of alkanes by Cl 2 or Br2 in the presence of ultraviolet light, as exemplified by the reactions of ethane (b) electrophilic addition of an alkene with a halogen, X2, or hydrogen halide, HX(g), at room temperature (c) substitution of an alcohol, e.g. by reaction with HX or KBr with H2SO4 or H3PO4; or with PCl 3 and heat; or with PCl 5; or with SOCl 2 2 classify halogenoalkanes into primary, secondary and tertiary 3 describe the following nucleophilic substitution reactions: (a) the reaction with NaOH(aq) and heat to produce an alcohol (b) the reaction with KCN in ethanol and heat to produce a nitrile (c) the reaction with NH3 in ethanol heated under pressure to produce an amine (d) the reaction with aqueous silver nitrate in ethanol as a method of identifying the halogen present as exemplified by bromoethane 15.1 Halogenoalkanes (continued) 4 describe the elimination reaction with NaOH in ethanol and heat to produce an alkene as exemplified by bromoethane 5 describe the SN1 and SN2 mechanisms of nucleophilic substitution in halogenoalkanes including the inductive effects of alkyl groups 6 recall that primary halogenoalkanes tend to react via the SN2 mechanism; tertiary halogenoalkanes via the SN1 mechanism; and secondary halogenoalkanes by a mixture of the two, depending on structure 7 describe and explain the different reactivities of halogenoalkanes (with particular reference to the relative strengths of the C–X bonds as exemplified by the reactions of halogenoalkanes with aqueous silver nitrates AS Chemistry: • AS Chemistry (New) A2 Chemistry: • A2 Chemistry (New) AS Physics: • AS Physics A2 Physics: • A2 Physics O level Physics: • O Level Physics NET Physics: • NET Physics AS Maths: • AS Mathematics O level Maths: • O Level Mathematics NET Maths: • NET Past Papers AS Business: • Плейлист A2 Business: • Плейлист AS Economics: • Плейлист A2 Economics: • Плейлист SAT Sample Papers: • SAT Mathematics Past Papers



















