REDOX (Reduction Oxidation) Reaction and Batteries (96) скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: REDOX (Reduction Oxidation) Reaction and Batteries (96) в качестве 4k
У нас вы можете посмотреть бесплатно REDOX (Reduction Oxidation) Reaction and Batteries (96) или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон REDOX (Reduction Oxidation) Reaction and Batteries (96) в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
REDOX (Reduction Oxidation) Reaction and Batteries (96)
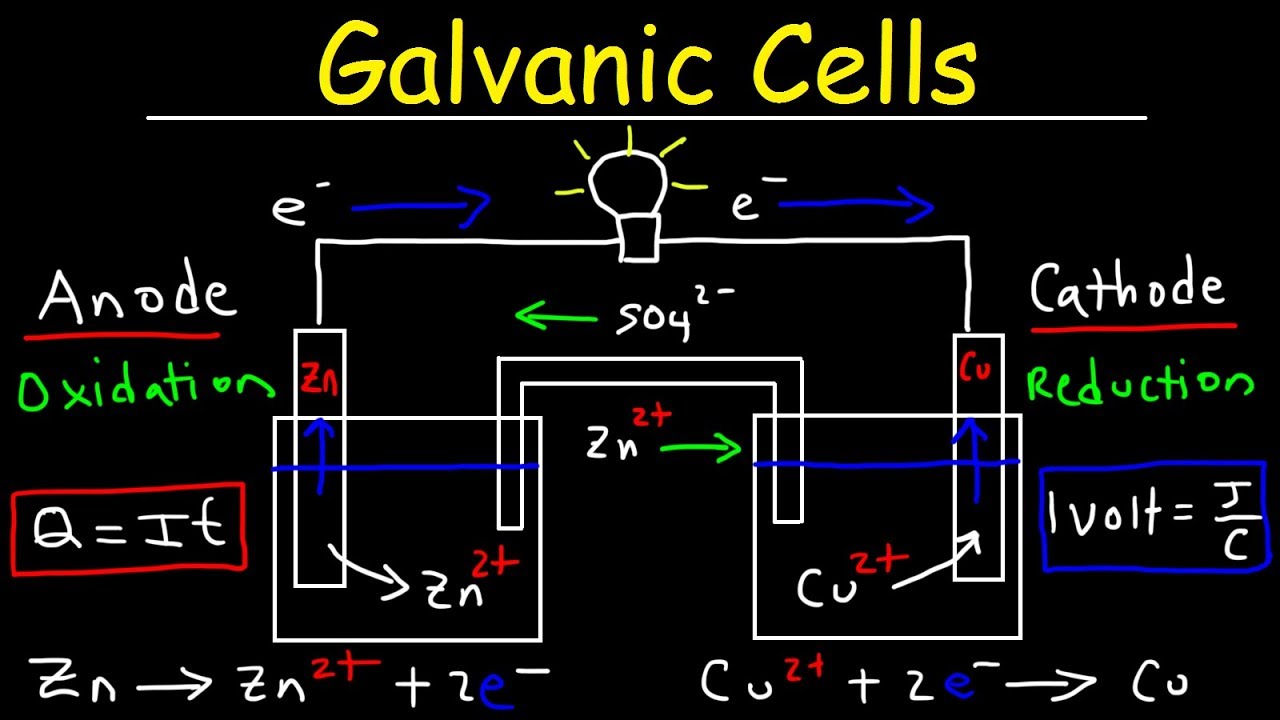
Electrochemistry is all about electrons---where they are and how we can harness their power! If we give electrons a path to happiness, which is what we do in a battery or a wire, then we can also force those electrons to travel where we want them to --- this IS electricity! All metals contain electrons, but electrons prefer to be on some metals rather than others, so by giving electrons the ability to get to a metal that they prefer, we can use their movement to our advantage (ie – a battery!) In a Galvanic Cell, electrons will always flow (or travel) from anode to cathode. The anode will end up losing mass while the cathode will end up gaining mass! This is because the electrons (an intrinsically negatively charged species) cause a change in charges. While the cathode becomes more and more negative as electrons flow to it, the cations in the solution begin to plate onto the Cathode, thus causing it to gain mass! When electrons transfer from one metal to another, we end up with a chemical reaction called REDOX, where charges change (due to the loss or gaining of electrons). It’s easiest to remember which metal does what by OIL RIG (Oxidation is Losing electrons, and Reduction is Gaining electrons). 🔔 SUBSCRIBE for more free education 🔔 ---------------------------------------------------------------- https://www.youtube.com/c/Duckonta?su... ---------------------------------------------------------------- 🦆 JOIN THE FLOCK 🦆 ---------------------------------------------------------------- • Facebook: / duckonta • Patreon: / duckonta ---------------------------------------------------------------- 💼 BUSINESS INQUIRIES 💼 ---------------------------------------------------------------- Duckonta@gmail.com ---------------------------------------------------------------- Thanks for watching! 0:00 Introduction 0:28 How do Batteries Work? 6:36 Which side of a battery would a Cation flow to? 7:56 Draw arrows to show the direction the electrons are flowing 9:23 Galvanic Cell 11:10 Salt Bridge of a Galvanic Cell 12:01 Oxidation is Losing, Reduction Is Gaining (OIL RIG) 13:16 Summary 14:30 Test Yourself Lead storage battery Redox reactions and electrochemistry Chemistry khan academy, Introduction to Oxidation Reduction (Redox) Reactions tyler dewitt, Oxidation and Reduction (Redox) Reactions Step-by-Step Example melissa maribel, Redox Reactions bozeman science, How To Balance Redox Reactions - General Chemistry Practice Test / Exam Review the organic chemistry tutor, Oxidation and Reduction Reactions - Basic Introduction the organic chemistry tutor, reduction oxidation wayne breslyn, Electrochemistry: Crash Course Chemistry Crashcourse, Redox Reactions: Crash Course Chemistry crashcourse, professor dave explains, reduction oxidation Chemistnate, reduction oxidation Learnohub, reduction oxidation Class 11, Additional Lecture 2. The Chemistry of Batteries (Intro to Solid-State Chemistry 2019) MIT opencourseware, What's inside Sumsung Battery? Mr. ax the backbencher experiment, How batteries work - Adam Jacobson ted-ed




![Пожалуй, главное заблуждение об электричестве [Veritasium]](https://imager.clipsaver.ru/6Hv2GLtnf2c/max.jpg)