Solving & Balancing Half Reaction REDOX Equations (98) скачать в хорошем качестве
Повторяем попытку...
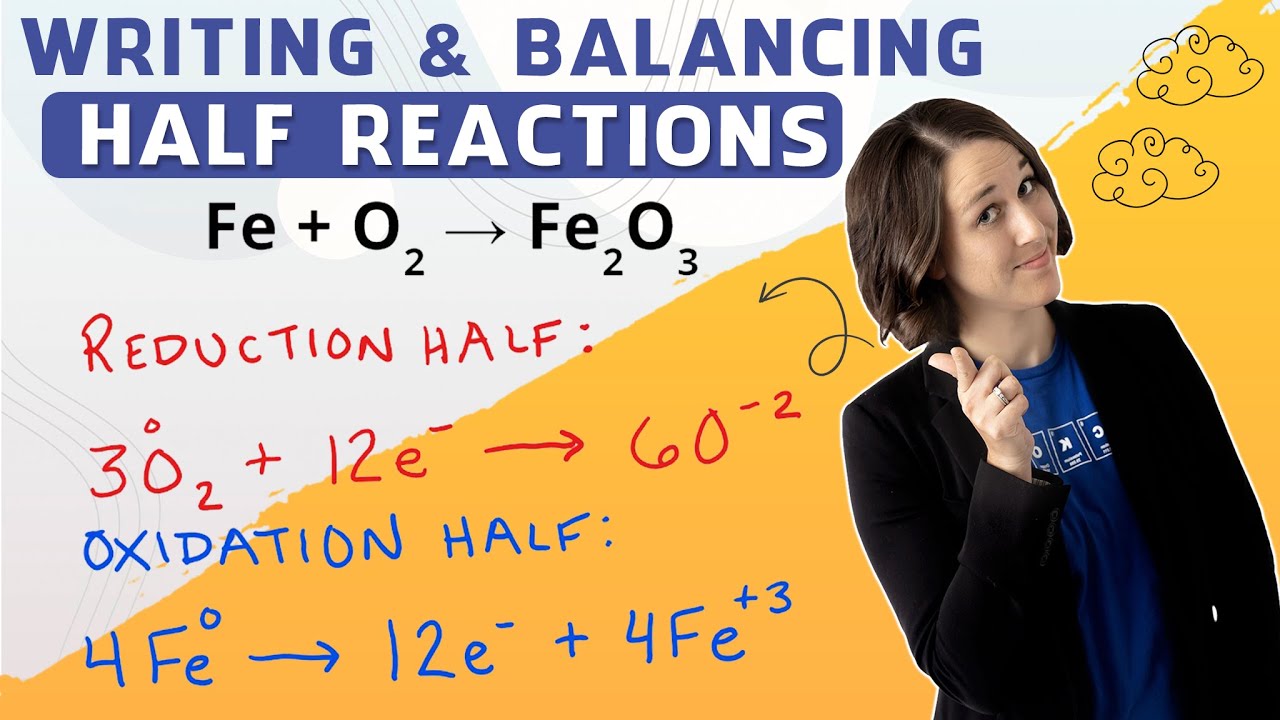
Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Solving & Balancing Half Reaction REDOX Equations (98) в качестве 4k
У нас вы можете посмотреть бесплатно Solving & Balancing Half Reaction REDOX Equations (98) или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Solving & Balancing Half Reaction REDOX Equations (98) в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Solving & Balancing Half Reaction REDOX Equations (98)
In order to be able to calculate cell potential (the actual voltage that could be produced from a REDOX reaction), a student first needs to know how to write and balance half reactions. Half reactions are just what they sound like: splitting a single REDOX reaction into its reduction and oxidation halves. This better demonstrates how many electrons are being gained and lost overall, and paints a clearer picture of what is happening at each electrode in a Galvanic Cell. In order to balance half reactions, we need to keep in mind that we are dealing with charges and electrons---so ionic compound subscripts and molar coefficients are trivial values here and only get in our way most of the time! What is necessary to focus on is if you have a diatomic species participating in the REDOX reaction, that you account for the number of atoms present (which WILL affect the end product charge!) This video walks you through how to write out each separate half-reaction, and then balance each to yield the overall electrochemical “reaction” in terms of the charged species participating. One of the examples is a classic challenge that many chemistry students struggle with: writing and balancing the half reaction for: 4Fe + 3O2 2Fe2O3 and also shows how to draw/model a Galvanic Cell using these half reactions. Take a quack at half reactions, you’ll be a pro in no time! 🔔 SUBSCRIBE for more free education 🔔 ---------------------------------------------------------------- https://www.youtube.com/c/Duckonta?su... ---------------------------------------------------------------- 🦆 JOIN THE FLOCK 🦆 ---------------------------------------------------------------- • Facebook: / duckonta • Patreon: / duckonta ---------------------------------------------------------------- 💼 BUSINESS INQUIRIES 💼 ---------------------------------------------------------------- Duckonta@gmail.com ---------------------------------------------------------------- Thanks for watching! 0:00 Introduction 0:16 Purpose of Half Reactions 0:58 Example 1 - Write the Reduction and Oxidation Half (Step by Step) 6:39 Example 2 - Write the Half Reaction for the REDOX Reaction (Step by Step) 11:27 Problem 3 - Write the Half Reaction for the Reduction/Oxidation Reaction (Test Yourself) 13:14 Problem 4 - Write the Half Reaction for the REDOX Reaction (Step by Step) 13:53 Modeling a Galvanic Cell 18:05 Summary 20:35 Test Yourself REDOX half reaction khan academy, REDOX half reaction tyler dewitt, REDOX half reaction melissa maribel, REDOX half reaction bozeman science, Half Reaction Method Balancing Redox Reactions In Basic Acidic Solution Chemistry the organic chemistry tutor, Standard Reduction Potentials of Half Reactions - Electrochemistry the organic chemistry tutor, REDOX half reaction wayne breslyn, REDOX half reaction Crashcourse, REDOX half reaction professor dave explains, What are the Half-Reactions? Chemistnate, Half Reaction Method jfr science, redox half reaction Learnohub, Chemistry 13.4 Writing Half-reactions for Redox isaacsteach, redox half reaction Class 11














![Пожалуй, главное заблуждение об электричестве [Veritasium]](https://imager.clipsaver.ru/6Hv2GLtnf2c/max.jpg)




