Residues vs. amino acids скачать в хорошем качестве
Повторяем попытку...

Скачать видео с ютуб по ссылке или смотреть без блокировок на сайте: Residues vs. amino acids в качестве 4k
У нас вы можете посмотреть бесплатно Residues vs. amino acids или скачать в максимальном доступном качестве, видео которое было загружено на ютуб. Для загрузки выберите вариант из формы ниже:
-
Информация по загрузке:
Скачать mp3 с ютуба отдельным файлом. Бесплатный рингтон Residues vs. amino acids в формате MP3:
Если кнопки скачивания не
загрузились
НАЖМИТЕ ЗДЕСЬ или обновите страницу
Если возникают проблемы со скачиванием видео, пожалуйста напишите в поддержку по адресу внизу
страницы.
Спасибо за использование сервиса ClipSaver.ru
Residues vs. amino acids
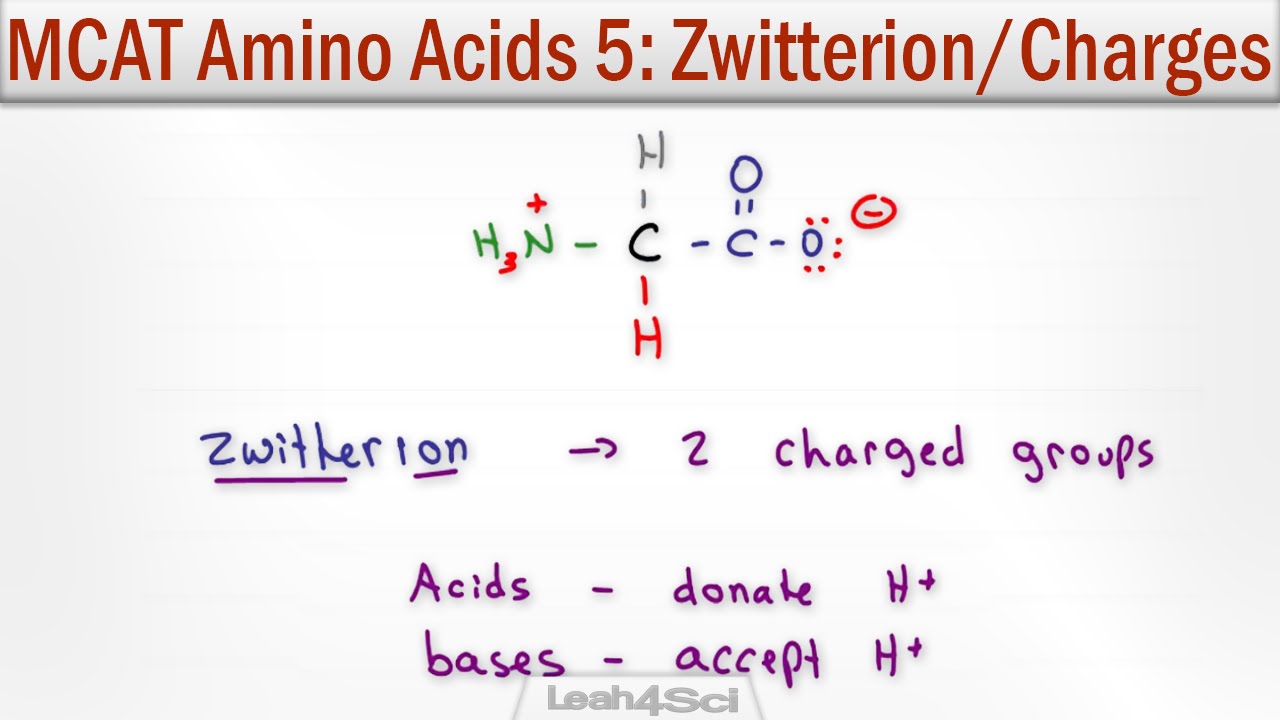
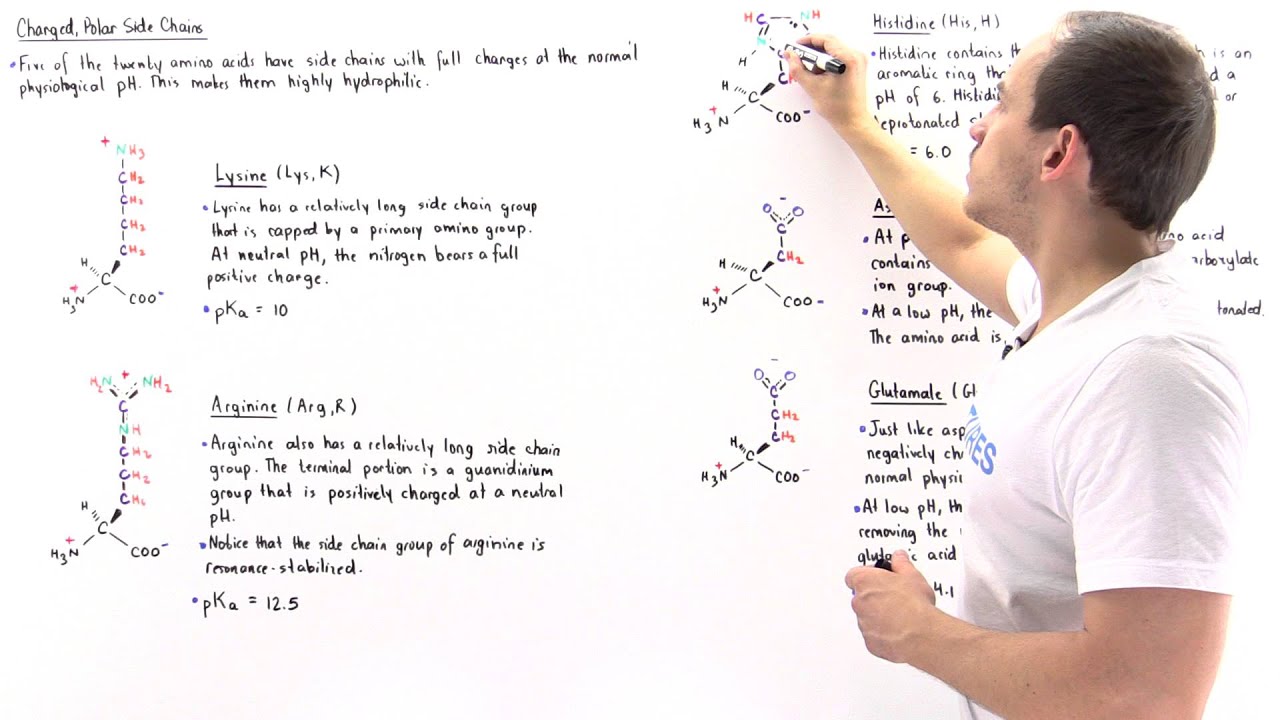
A “residue” is ‘just’ a word we use for what used to be called an amino acid once it’s in the context of a peptide or protein. But it’s more than (and physically less than!) “just” an amino acid. Instead, the properties of the side chain can be dramatically different than its properties in a free-floating amino acid. This allows for cool things like lysines and serines only becoming strongly nucleophilic (and thus reactive) when they’re residues in specific contexts, etc. Let me back up and explain… glossary entry: https://thebumblingbiochemist.com/glo... note: text old, video new First off, what is an amino acid anyway? Like many (but not all) things in science, hints are in the name. At the core is the central (alpha) carbon (Cα) - which is hooked up to the unique charm (side chain or “R group”) which gives different amino acids their special “superpowers” and the generic parts that allow for linking and make it an amino acid - “Amino” refers to them having an “amine” group - a nitrogen (N) hooked up to hydrogen(s) (H) and/or carbon(s) (C). And “acid” refers to them having a carboxylic acid group - a C double-bonded to an oxygen (O) and also bonded to a hydroxyl (-OH) group (so (-(C=O)-OH). An “acid” (in one definition) is something that donates a proton (an H⁺), and a carboxylic acid can donate a proton from the hydroxyl group to give you a carboxylate anion (-(C=O)-O⁻). The neutral form of the amine group (-NH₂) can act as a base (proton taker) to become the cationic -NH₃⁺. Note: an “ion” is just a name for any type of charged particle - we call something an “anion” if it has a negative charge (which comes from having more electrons than protons) and we call something a “cation” if it has a positive charge (comes from having more protons than electrons). Protons and electrons are oppositely-charged “subatomic particles” that make up atoms Which protonation state these end groups are in depends on the pH (a measure of how many free protons are floating around). At physiological (bodily) pH (~7.4) the “zwitterionic” form is the most common - I love this word and it just means that you have a positive charged group (like -NH₃⁺) and a negative charged group (-(C=O)-O⁻) in the same molecule, so the charges cancel out to give you a neutral molecule overall. Sorry if this is too technical for your hearts’ desires, but I don’t want you to get thrown off if you see amino acids written and/or drawn out with different protonation states. much more on this here: https://bit.ly/zwitterions & • The zwitterionic life of amino acids But turns out that, once amino acids link up, you only have 2 such ends to worry about… “Amino acids” are the “free-floating” forms of protein letters - when they link together, they do so by joining the carbonyl (C=O) carbon of one amino acid to the nitrogen of the amino group of the other amino acid, physically losing the equivalent of water (2 H & 1 O) in the process - and also “functionally” losing the free amino and carboxyl groups that are now merged into a peptide bond. As a result, when it comes to the generic peptide backbone, only the “first” amino acid will have a free amino group (we call this the “N terminus” and only the “last” amino acid will have a free carboxylic acid group (we call this the “C terminus”) So, instead of calling them “amino acids,” once they’re linked together and no longer “amino acids” we call the “residuals” of what used to be individual amino acids “residues” (sorry if this is too technical, but this confused me for the longest time when I was an undergrad and I was embarrassed to ask! (speaking of which - never be embarrassed to ask questions - not only does it hold you back intellectually, it’s way more embarrassing to find out years later you’ve got it all wrong). The distinction can matter because the side chains may act slightly differently (e.g be more/less acidic or more/less nucleophilic) in the context of a peptide or protein vs when it’s as a free-floating amino acid. This is why, for example, the side chains in the active sites of enzymes might be more reactive than you’d expect. Because all those contexts are different, there will be a range of reactivities, etc. for the “same” residue and using the term “residue” has the added benefit of reminding us that we may need to consider the context! Much more on amino acids: http://bit.ly/allaminoacids ; YouTube: • Amino acids - all about 'em! more about all sorts of things: #365DaysOfScience All (with topics listed) 👉 http://bit.ly/2OllAB0 or search blog: http://thebumblingbiochemist.com